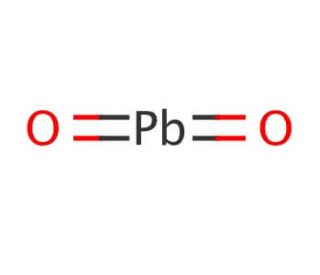
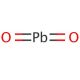
Molecular structure of Lead(IV) oxide, CAS Number: 1309-60-0
Lead(IV) oxide (CAS 1309-60-0)
Alternate Names:
Lead peroxide
CAS Number:
1309-60-0
Purity:
≥97%
Molecular Weight:
239.20
Molecular Formula:
PbO2
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Lead(IV) oxide, a potent oxidizer, finds utility in multiple electrochemical applications. In the field of electrochemical sensing, it serves as an electrode material, enabling the detection of diverse analytes like glucose, lactate, and amino acids. In the realm of electrochemical energy storage, Lead(IV) oxide acts as a cathode material, contributing to the efficient functioning of lithium-ion batteries. Furthermore, in electrochemical synthesis, it assumes the role of a catalyst, facilitating the oxidation of organic compounds.
Lead(IV) oxide (CAS 1309-60-0) References
- Screening and confirmation of triphenylmethane dyes and their leuco metabolites in trout muscle using HPLC-vis and ESP-LC-MS. | Tarbin, JA., et al. 1998. Analyst. 123: 2567-71. PMID: 10435301
- Raman spectroscopy as a means for the identification of plattnerite (PbO2), of lead pigments and of their degradation products. | Burgio, L., et al. 2001. Analyst. 126: 222-7. PMID: 11235107
- PbO2-based graphite-epoxy electrode for potentiometric determination of acids and bases in aqueous and aqueous-ethanolic media. | Teixeira, MF., et al. 2001. Fresenius J Anal Chem. 370: 383-6. PMID: 11495060
- Lead(IV) oxide-graphite composite electrodes: application to sensing of ammonia, nitrite and phenols. | Sljukić, B., et al. 2007. Anal Chim Acta. 587: 240-6. PMID: 17386779
- Effects of pH and carbonate concentration on dissolution rates of the lead corrosion product PbO(2). | Xie, Y., et al. 2010. Environ Sci Technol. 44: 1093-9. PMID: 20063875
- Formation of lead(IV) oxides from lead(II) compounds. | Wang, Y., et al. 2010. Environ Sci Technol. 44: 8950-6. PMID: 21047060
- Kinetics of the reductive dissolution of lead(IV) oxide by iodide. | Wang, Y., et al. 2012. Environ Sci Technol. 46: 5859-66. PMID: 22540764
- Kinetics of lead(IV) oxide (PbO2) reductive dissolution: role of lead(II) adsorption and surface speciation. | Wang, Y., et al. 2013. J Colloid Interface Sci. 389: 236-43. PMID: 23062963
- Diastereoselective synthesis of 5-hydroxy-8-methoxy-1-oxaspiro[5,5]undeca-7,10-diene-9-one. | Plourde, GL. and Scully, TW. 2013. Molecules. 18: 1174-80. PMID: 23344206
- Redox-Driven Recrystallization of PbO2. | Pan, W., et al. 2022. Environ Sci Technol. 56: 7864-7872. PMID: 35654758
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Lead(IV) oxide, 100 g | sc-211730 | 100 g | $125.00 | |||
Lead(IV) oxide, 500 g | sc-211730A | 500 g | $290.00 |
