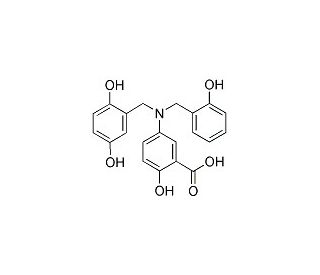
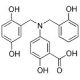
Molecular structure of Lavendustin A, CAS Number: 125697-92-9
Lavendustin A (CAS 125697-92-9)
Alternate Names:
5-Amino-[(N-2,5-dihydroxybenzyl)-N′-2-hydroxybenzyl]salicylic acid;RG14355
Application:
Lavendustin A is a selective and potent inhibitor of the receptor tyrosine kinase
CAS Number:
125697-92-9
Purity:
≥95%
Molecular Weight:
381.38
Molecular Formula:
C21H19NO6
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Lavendustin A selectively and potently inhibits EGFR (epidermal growth factor receptor) tyrosine kinase. The chemical has been observed to bind to a site on the kinase that is different from the peptide substrate and ATP binding sites. Lavendustin A is an inhibitor of c-Src.
Lavendustin A (CAS 125697-92-9) References
- Inhibition of atrial wall stretch-induced cardiac hormone secretion by lavendustin A, a potent tyrosine kinase inhibitor. | Taskinen, P., et al. 1999. Endocrinology. 140: 4198-207. PMID: 10465292
- Protein tyrosine kinase activity of lavendustin A and the phytoestrogen genistein on progesterone synthesis in cultured rat ovarian cells. | Whitehead, SA. and Lacey, M. 2000. Fertil Steril. 73: 613-9. PMID: 10689022
- Acute and chronic effects of genistein, tyrphostin and lavendustin A on steroid synthesis in luteinized human granulosa cells. | Whitehead, SA., et al. 2002. Hum Reprod. 17: 589-94. PMID: 11870108
- Synthesis and investigation of conformationally restricted analogues of lavendustin A as cytotoxic inhibitors of tubulin polymerization. | Mu, F., et al. 2002. J Med Chem. 45: 4774-85. PMID: 12361405
- Structure-activity relationship studies on a novel class of antiproliferative agents derived from Lavendustin A. Part I: Ring A modifications. | Nussbaumer, P. and Winiski, AP. 2008. Bioorg Med Chem. 16: 7552-60. PMID: 18678497
- Inhibition of tyrosine kinase and epidermal growth factor receptor internalization by lavendustin A methyl ester in cultured A431 cells. | Onoda, T., et al. 1990. Drugs Exp Clin Res. 16: 249-53. PMID: 2086161
- Isolation of a novel tyrosine kinase inhibitor, lavendustin A, from Streptomyces griseolavendus. | Onoda, T., et al. 1989. J Nat Prod. 52: 1252-7. PMID: 2614420
- Suppression of VEGF-induced angiogenesis by the protein tyrosine kinase inhibitor, lavendustin A. | Hu, DE. and Fan, TP. 1995. Br J Pharmacol. 114: 262-8. PMID: 7533611
- Novel antiproliferative agents derived from lavendustin A. | Nussbaumer, P., et al. 1994. J Med Chem. 37: 4079-84. PMID: 7990108
- Non-amine based analogues of lavendustin A as protein-tyrosine kinase inhibitors. | Smyth, MS., et al. 1993. J Med Chem. 36: 3010-4. PMID: 8105084
- Tyrosine kinase inhibitors, herbimycin A and lavendustin A, block formation of long-term potentiation in the dentate gyrus in vivo. | Abe, K. and Saito, H. 1993. Brain Res. 621: 167-70. PMID: 8221070
- SDZ 281-977: a modified partial structure of lavendustin A that exerts potent and selective antiproliferative activities in vitro and in vivo. | Cammisuli, S., et al. 1996. Int J Cancer. 65: 351-9. PMID: 8575857
- Chloride efflux during the progesterone-initiated human sperm acrosome reaction is inhibited by lavendustin A, a tyrosine kinase inhibitor. | Meizel, S. and Turner, KO. 1996. J Androl. 17: 327-30. PMID: 8889693
Inhibitor of:
c-Src, CABYR, CASKIN2, CD45RC, EGFR, LOC147710, LOC388259, OTTMUSG00000015806, OTTMUSG00000015960, OTTMUSG00000016203, OTTMUSG00000016254, Sad-1, SSH2, Synapsin IIIa, and TESK1.Activator of:
p35, PID1, and Plb2.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Lavendustin A, 1 mg | sc-200539 | 1 mg | $116.00 | |||
Lavendustin A, 5 mg | sc-200539A | 5 mg | $456.00 |
