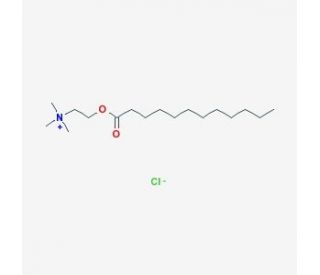
Lauroylcholine Chloride (CAS 25234-60-0)
QUICK LINKS
Lauroylcholine Chloride, with CAS number 25234-60-0, is a quaternary ammonium salt where the typical choline molecule is esterified with a twelve-carbon lauric acid, resulting in a compound that combines lipid-like and choline-like properties. This chemical structure enables Lauroylcholine Chloride to interact uniquely with biological membranes and various surfactant systems, making it an interesting subject for research in biochemistry and materials sciences. In research contexts, this compound has been primarily used to study the interaction between quaternary ammonium compounds and lipid bilayers, as well as their potential to modify membrane properties such as permeability and fluidity. Its amphiphilic nature allows Lauroylcholine Chloride to integrate into lipid layers, affecting the packing and overall stability of these structures. This has implications for understanding the mechanism of action of surfactants and their applications in formulating products where control of lipid membrane behavior is crucial. Additionally, the compound′s ability to form stable micelles and other aggregate structures in aqueous solutions makes it a valuable tool in the study of micellar systems, where its effects on solubilization and emulsification are examined. These characteristics are critical for advancing knowledge in areas such as formulation chemistry and the development of delivery systems in non-medical fields.
Lauroylcholine Chloride (CAS 25234-60-0) References
- Isolation and properties of the luciferase stored in the ovary of the scyphozoan medusa Periphylla periphylla. | Shimomura, O., et al. 2001. Biol Bull. 201: 339-47. PMID: 11751246
- Nanoparticles from lipid-based liquid crystals: emulsifier influence on morphology and cytotoxicity. | Murgia, S., et al. 2010. J Phys Chem B. 114: 3518-25. PMID: 20170140
- Biotransformation of alkanoylcholines under methanogenic conditions. | Watson, MK., et al. 2012. Water Res. 46: 2947-56. PMID: 22463863
- Physicochemical, cytotoxic, and dermal release features of a novel cationic liposome nanocarrier. | Carboni, M., et al. 2013. Adv Healthc Mater. 2: 692-701. PMID: 23184424
- Enhancing the biodegradation of oil in sandy sediments with choline: a naturally methylated nitrogen compound. | Mortazavi, B., et al. 2013. Environ Pollut. 182: 53-62. PMID: 23896678
- Thermoluminescence investigations on the site of action of o-phthalaldehyde in photosynthetic electron transport. | Desai, TS., et al. 1996. Photosynth Res. 48: 213-20. PMID: 24271301
- Photoproduction of hydrogen peroxide in Photosystem II membrane fragments: A comparison of four signals. | Klimov, V., et al. 1993. Photosynth Res. 38: 409-16. PMID: 24317997
- 3-hydroxycoumarin loaded vesicles for recombinant human tyrosinase inhibition in topical applications. | Schlich, M., et al. 2018. Colloids Surf B Biointerfaces. 171: 675-681. PMID: 30107341
- The effect of absorption enhancers on the oral absorption of the GP IIB/IIIA receptor antagonist, DMP 728, in rats and dogs. | Burcham, DL., et al. 1995. Pharm Res. 12: 2065-70. PMID: 8786991
- The effect of DNA chain length and chemical structure of the surfactant molecule on the properties of the DNA-based bio-organic field effect transistor memory. | Liang, Li Juan, et al. 2012. 한국화상학회지. 18.3: 16-24.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Lauroylcholine Chloride, 5 g | sc-211723 | 5 g | $360.00 |
