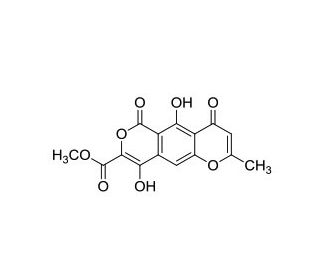
Lateropyrone (CAS 93752-78-4)
QUICK LINKS
Lateropyrone, a natural product derived from fungal sources, has emerged as a significant compound in scientific research due to its intriguing chemical structure and promising biological activities. Recent studies have explaind its mechanism of action, revealing that lateropyrone exerts its effects by modulating cellular signaling pathways involved in various physiological processes. Specifically, lateropyrone has been found to interact with protein kinases, particularly those involved in cellular proliferation, differentiation, and survival. This interaction results in the regulation of downstream signaling cascades, leading to alterations in gene expression and cellular behavior. Furthermore, research has demonstrated that lateropyrone possesses potent anti-inflammatory properties, with the ability to inhibit the production of pro-inflammatory mediators and attenuate inflammatory responses in vitro and in vivo. Additionally, lateropyrone has shown promise as a potential antitumor agent, with studies indicating its ability to induce apoptosis and inhibit cancer cell proliferation in various cancer models. Moreover, its unique chemical structure and biological activities have prompted further investigations into its pharmacological potential and medicinal chemistry, with the aim of developing novel agents targeting inflammatory diseases, cancer, and other health conditions.
Lateropyrone (CAS 93752-78-4) References
- Multiple regression analysis as a tool for the identification of relations between semi-quantitative LC-MS data and cytotoxicity of extracts of the fungus Fusarium avenaceum (syn. F. arthrosporioides). | Uhlig, S., et al. 2006. Toxicon. 48: 567-79. PMID: 16908037
- Inducing secondary metabolite production by the endophytic fungus Fusarium tricinctum through coculture with Bacillus subtilis. | Ola, AR., et al. 2013. J Nat Prod. 76: 2094-9. PMID: 24175613
- Co-cultivation--a powerful emerging tool for enhancing the chemical diversity of microorganisms. | Marmann, A., et al. 2014. Mar Drugs. 12: 1043-65. PMID: 24549204
- Elicitation of secondary metabolism in actinomycetes. | Abdelmohsen, UR., et al. 2015. Biotechnol Adv. 33: 798-811. PMID: 26087412
- Bioprospecting endophytic fungi from Fusarium genus as sources of bioactive metabolites. | Toghueo, RMK. 2020. Mycology. 11: 1-21. PMID: 32128278
- Co-culture of the fungus Fusarium tricinctum with Streptomyces lividans induces production of cryptic naphthoquinone dimers. | Moussa, M., et al. 2019. RSC Adv. 9: 1491-1500. PMID: 35518011
- Structural elucidation of an antibiotic from the fungus Fusarium avenaceum Fries Sacc.; an amended structure for lateropyrone. | Gorst-Allman and C. P., et al. 1986. S. Afr. J. Chem.;(South Africa). 39.2: 116-117.
- Antibiotic mycotoxins from an endophytic Fusarium acuminatum isolated from the medicinal plant Geum macrophyllum. | Clark and Trevor N., et al. 2018. Natural Product Communications. 13.10: 1934578X1801301017.
- Co-culture of the bacterium Pseudomonas aeruginosa with the fungus Fusarium tricinctum induces bacterial antifungal and quorum sensing signaling molecules. | Moussa and Mariam, et al. 2020. Phytochemistry letters. 36: 37-41.
- Fusaristatins D–F and (7S, 8R)-(−)-chlamydospordiol from Fusarium sp. BZCB-CA, an endophyte of Bothriospermum chinense. | Ariantari and Ni Putu, et al. 2021. Tetrahedron. 85: 132065.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Lateropyrone, 5 mg | sc-202206 | 5 mg | $147.00 |
