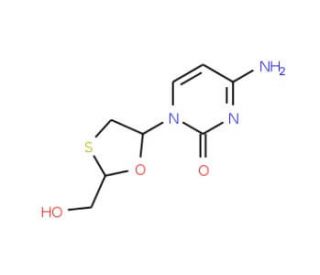
Lamivudine-15N2,13C
QUICK LINKS
Lamivudine-15N2,13C is a stable isotope-labeled variant of lamivudine, where specific nitrogen atoms are replaced with nitrogen-15 (15N) and certain carbon atoms with carbon-13 (13C). This isotopic labeling is extensively used in research to enhance the precision of analytical techniques, such as mass spectrometry and nuclear magnetic resonance (NMR) spectroscopy, facilitating the study of the compound′s metabolic pathways and interactions within biological systems. Lamivudine is a nucleoside analog reverse transcriptase inhibitor (NRTI), which is incorporated into viral DNA by viral reverse transcriptase, leading to chain termination. It mimics the natural nucleoside cytidine but, once phosphorylated and incorporated into the DNA, it lacks the 3′ hydroxyl group necessary for the formation of the next phosphodiester bond in the DNA backbone, thus terminating DNA synthesis. This mechanism is crucial for understanding how nucleoside analogs inhibit viral replication. In research, Lamivudine-15N2,13C is particularly useful for detailed pharmacokinetic and pharmacodynamic studies. The isotopic labels allow for the tracing of lamivudine through various metabolic processes without interference from other biological molecules, enabling researchers to study how the compound is absorbed, distributed, metabolized, and excreted with high accuracy. Furthermore, Lamivudine-15N2,13C is used to investigate the enzyme-substrate interactions involved in the activation and incorporation of lamivudine into viral DNA. By using this labeled variant, scientists can observe subtle changes in how the compound interacts with viral reverse transcriptase and host cell kinases, which are responsible for phosphorylating the nucleoside analog into its active triphosphate form.
Lamivudine-15N2,13C References
- Lamivudine Concentration in Hair and Prediction of Virologic Failure and Drug Resistance among HIV Patients Receiving Free ART in China. | Yan, J., et al. 2016. PLoS One. 11: e0154421. PMID: 27119346
- Identifying Risk Factors for Recent HIV Infection in Kenya Using a Recent Infection Testing Algorithm: Results from a Nationally Representative Population-Based Survey. | Kim, AA., et al. 2016. PLoS One. 11: e0155498. PMID: 27195800
- Development, validation and clinical application of a method for the simultaneous quantification of lamivudine, emtricitabine and tenofovir in dried blood and dried breast milk spots using LC-MS/MS. | Waitt, C., et al. 2017. J Chromatogr B Analyt Technol Biomed Life Sci. 1060: 300-307. PMID: 28651173
- Population pharmacokinetics of abacavir and lamivudine in severely malnourished human immunodeficiency virus-infected children in relation to treatment outcomes. | Archary, M., et al. 2019. Br J Clin Pharmacol. 85: 2066-2075. PMID: 31141195
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Lamivudine-15N2,13C, 1 mg | sc-280894 | 1 mg | $533.00 |
