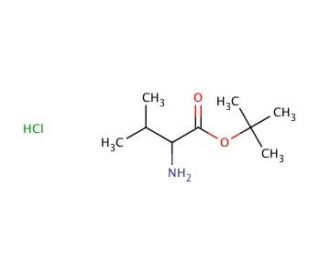
L-Valine tert-butyl ester hydrochloride (CAS 13518-40-6)
QUICK LINKS
L-Valine tert-butyl ester hydrochloride (L-VTBH) serves as a synthetic variant of the essential amino acid L-valine. Its applications span a wide range, with prominent uses as a reagent in peptide synthesis, a fundamental constituent for peptide libraries, and a substrate for enzymatic reactions. L-Valine tert-butyl ester hydrochloride finds extensive utilization across diverse scientific research applications. It has been instrumental in peptide synthesis, serving as a key building block for constructing peptide libraries, as well as functioning as a substrate for enzymatic reactions. Notably, it has been employed to explore the effects of L-valine on cellular metabolism, protein synthesis, and enzyme activity.
L-Valine tert-butyl ester hydrochloride (CAS 13518-40-6) References
- Enzyme-labile protecting groups in peptide synthesis: development of glucose- and galactose-derived urethanes. | Gum, AG., et al. 2000. Chemistry. 6: 3714-21. PMID: 11073241
- Synthesis and characterization of peptides containing a cyclic Val adduct of diepoxybutane, a possible biomarker of human exposure to butadiene. | Jayaraj, K., et al. 2003. Chem Res Toxicol. 16: 637-43. PMID: 12755593
- Fluorous (trimethylsilyl)ethanol: a new reagent for carboxylic acid tagging and protection in peptide synthesis. | Fustero, S., et al. 2006. J Org Chem. 71: 3299-302. PMID: 16599635
- Discovery of imidazolidine-2,4-dione-linked HIV protease inhibitors with activity against lopinavir-resistant mutant HIV. | Flosi, WJ., et al. 2006. Bioorg Med Chem. 14: 6695-712. PMID: 16828558
- Total synthesis of syringolin A and B. | Pirrung, MC., et al. 2010. Org Lett. 12: 2402-5. PMID: 20426399
- Investigation of the synergistic effect with amino acid-derived chiral ionic liquids as additives for enantiomeric separation in capillary electrophoresis. | Zhang, J., et al. 2013. J Chromatogr A. 1316: 119-26. PMID: 24119759
- Evaluation of vancomycin-based synergistic system with amino acid ester chiral ionic liquids as additives for enantioseparation of non-steroidal anti-inflammatory drugs by capillary electrophoresis. | Zhang, J., et al. 2014. Talanta. 119: 193-201. PMID: 24401404
- C-terminal peptide extension via gas-phase ion/ion reactions. | Peng, Z. and McLuckey, SA. 2015. Int J Mass Spectrom. 391: 17-23. PMID: 26640400
- Capillary electrophoresis separation of phenethylamine enantiomers using amino acid based ionic liquids. | Wahl, J. and Holzgrabe, U. 2018. J Pharm Biomed Anal. 148: 245-250. PMID: 29059613
- Non-radical synthesis of chitosan-quercetin polysaccharide: Properties, bioactivity and applications. | Shebis, Y., et al. 2022. Carbohydr Polym. 284: 119206. PMID: 35287917
- Synthesis of reagents for the construction of hypusine and deoxyhypusine peptides and their application as peptidic antigens. | Bergeron, RJ., et al. 1998. J Med Chem. 41: 3888-900. PMID: 9748364
- Synthesis of Sulfonimidamide-Based Amino Acid Building Blocks with Orthogonal Protecting Groups | Praveen K. Chinthakindi, Andrea Benediktsdottir, Ayah Ibrahim, Atta Wared, Carl-Johan Aurell, Anna Pettersen, Edouard Zamaratski, Per I. Arvidsson, Yantao Chen, Anja Sandström. February 07, 2019. European Journal of Organic Chemistry. Volume2019, Issue5: Pages 1045-1057.
- Novel highly-soluble peptide–chitosan polymers: Chemical synthesis and spectral characterization | M.K.S. Batista a b, L.F. Pinto a b, C.A.R. Gomes a, P. Gomes b. 11 May 2006,. Carbohydrate Polymers. Volume 64, Issue 2,: Pages 299-305.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
L-Valine tert-butyl ester hydrochloride, 5 g | sc-228402 | 5 g | $159.00 |
