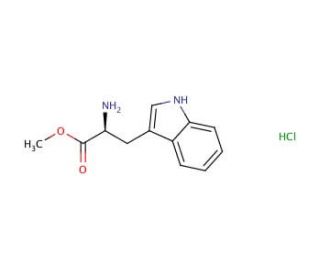
L-Tryptophan Methyl Ester Hydrochloride (CAS 7524-52-9)
QUICK LINKS
L-Tryptophan Methyl Ester Hydrochloride is frequently employed in biochemical research, particularly in studies focused on peptide synthesis and protein interactions. L-Tryptophan Methyl Ester Hydrochloride, a derivative of the amino acid L-tryptophan, is used to investigate how modifications to amino acids can affect peptide structure and function. L-Tryptophan Methyl Ester Hydrochloride is utilized to explore its incorporation into peptides, which aids in understanding how such modifications influence biological activity and stability of peptide-based compounds. L-Tryptophan Methyl Ester Hydrochloride is useful in studies aimed at elucidating the mechanisms of protein folding and misfolding, which are key to understanding various biochemical processes. Its role in the synthesis of novel peptides with potential industrial applications is also a significant area of study, contributing to advancements in biotechnology and materials science.
L-Tryptophan Methyl Ester Hydrochloride (CAS 7524-52-9) References
- The Chiral Pool in the Pictet-Spengler Reaction for the Synthesis of β-Carbolines. | Dalpozzo, R. 2016. Molecules. 21: PMID: 27240334
- Furan- and Thiophene-2-Carbonyl Amino Acid Derivatives Activate Hypoxia-Inducible Factor via Inhibition of Factor Inhibiting Hypoxia-Inducible Factor-1. | Kawaguchi, SI., et al. 2018. Molecules. 23: PMID: 29641495
- Isolation and Synthesis of Misszrtine A: A Novel Indole Alkaloid From Marine Sponge-Associated Aspergillus sp. SCSIO XWS03F03. | Zhou, R., et al. 2018. Front Chem. 6: 212. PMID: 29951479
- Questioning the γ-gauche effect: stereoassignment of 1,3-disubstituted-tetrahydro-β-carbolines using 1H-1H coupling constants. | Cagašová, K., et al. 2019. Org Biomol Chem. 17: 6687-6698. PMID: 31232413
- Examination of sulfonamide-based inhibitors of MMP3 using the conditioned media of invasive glioma cells. | Poole, AT., et al. 2020. J Enzyme Inhib Med Chem. 35: 672-681. PMID: 32156166
- Role of Indole Scaffolds as Pharmacophores in the Development of Anti-Lung Cancer Agents. | Dhuguru, J. and Skouta, R. 2020. Molecules. 25: PMID: 32244744
- 3-Phenyllactic acid, a root-promoting substance isolated from Bokashi fertilizer, exhibits synergistic effects with tryptophan. | Maki, Y., et al. 2021. Plant Biotechnol (Tokyo). 38: 9-16. PMID: 34177319
- Development of a simple method for measuring tedizolid concentration in human serum using HPLC with a fluorescent detector. | Tsuji, Y., et al. 2021. Medicine (Baltimore). 100: e28127. PMID: 34889275
- Anthranilamides with quinoline and β-carboline scaffolds: design, synthesis, and biological activity. | Beus, M., et al. 2022. Mol Divers. 26: 2595-2612. PMID: 34997441
- A photochemical and theoretical study of the triplet reactivity of furano- and pyrano-1,4-naphthoquionones towards tyrosine and tryptophan derivatives. | Teixeira, RI., et al. 2019. RSC Adv. 9: 13386-13397. PMID: 35519567
- Design, synthesis and evaluation of inhibitors of the SARS-CoV-2 nsp3 macrodomain. | Sherrill, LM., et al. 2022. Bioorg Med Chem. 67: 116788. PMID: 35597097
- Total Synthesis of the Four Stereoisomers of Cyclo(l-Trp-l-Arg) Raises Uncertainty of the Structures of the Natural Products and Invalidates Their Promising Antimicrobial Activities. | Chen, D., et al. 2022. Molecules. 27: PMID: 36144649
- Facile Synthesis of Some Coumarin Derivatives and Their Cytotoxicity through VEGFR2 and Topoisomerase II Inhibition. | Gomaa, MS., et al. 2022. Molecules. 27: PMID: 36500372
- Cationic Surfactants Based on Arginine-Phenylalanine and Arginine-Tryptophan: Synthesis, Aggregation Behavior, Antimicrobial Activity, and Biodegradation. | Pérez, L., et al. 2022. Pharmaceutics. 14: PMID: 36559096
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
L-Tryptophan Methyl Ester Hydrochloride, 5 g | sc-391987 | 5 g | $34.00 | |||
L-Tryptophan Methyl Ester Hydrochloride, 25 g | sc-391987A | 25 g | $133.00 |
