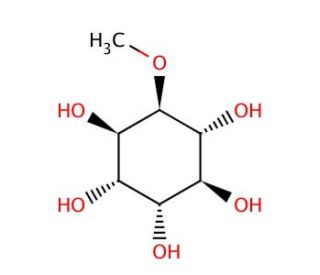
Molecular structure of L-Quebrachitol, CAS Number: 642-38-6
L-Quebrachitol (CAS 642-38-6)
Alternate Names:
2-O-Methyl-chiro-inositol
Application:
L-Quebrachitol is an important starting material for synthesis of optically active inositol phosphates
CAS Number:
642-38-6
Purity:
≥98%
Molecular Weight:
194.18
Molecular Formula:
C7H14O6
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
L-Quebrachitol, a naturally occurring cyclitol or methylated inositol, is distinguished by its pivotal role as a chiral precursor in the synthesis of optically active inositol phosphates. This compound, characterized by its unique structural configuration, includes a hydroxyl group at every carbon except for one, which is methylated, providing a stereocenter that is for its activity in synthetic pathways. Inositol phosphates play critical roles in cellular processes, including signal transduction, membrane trafficking, and ion channel regulation, making the synthesis of their optically active forms a significant endeavor in both chemical and biological research.
L-Quebrachitol (CAS 642-38-6) References
- A versatile approach to PI(3,4)P2, PI(4,5)P2, and PI(3,4,5)P3 from L-(-)-quebrachitol. | Qiao, L., et al. 2000. Org Lett. 2: 115-7. PMID: 10814260
- Functional role of polyhydroxy compounds on protein structure and thermal stability studied by circular dichroism spectroscopy. | Ortbauer, M. and Popp, M. 2008. Plant Physiol Biochem. 46: 428-34. PMID: 18343146
- First record of L-quebrachitol in Allophylus edulis (Sapindaceae). | Díaz, M., et al. 2008. Carbohydr Res. 343: 2699-700. PMID: 18715552
- Inositols and methylinositols in sea buckthorn (Hippophaë rhamnoides) berries. | Kallio, H., et al. 2009. J Chromatogr B Analyt Technol Biomed Life Sci. 877: 1426-32. PMID: 19345619
- Experimental and theoretical investigation of epoxide quebrachitol derivatives through spectroscopic analysis. | De Almeida, MV., et al. 2010. Org Lett. 12: 5458-61. PMID: 21043479
- (1) H NMR analysis of O-methyl-inositol isomers: a joint experimental and theoretical study. | De Almeida, MV., et al. 2012. Magn Reson Chem. 50: 608-14. PMID: 22865668
- Methyl-inositol, γ-aminobutyric acid and other health benefit compounds in the aril of litchi. | Wu, ZC., et al. 2016. Int J Food Sci Nutr. 67: 762-72. PMID: 27314889
- Chiral synthesis of D- and L-myo-inositol 1,4,5-trisphosphate. | Tegge, W. and Ballou, CE. 1989. Proc Natl Acad Sci U S A. 86: 94-8. PMID: 2783488
- Water Solvent Effect on Theoretical Evaluation of 1H NMR Chemical Shifts: o-Methyl-Inositol Isomer. | Dos Santos, HF., et al. 2017. J Phys Chem A. 121: 2839-2846. PMID: 28328211
- l-Quebrachitol Promotes the Proliferation, Differentiation, and Mineralization of MC3T3-E1 Cells: Involvement of the BMP-2/Runx2/MAPK/Wnt/β-Catenin Signaling Pathway. | Yodthong, T., et al. 2018. Molecules. 23: PMID: 30486330
- Phytotoxic and nematicide evaluation of Croton ehrenbergii (Euphorbiaceae). | Rios, MY., et al. 2019. Pest Manag Sci. 75: 2158-2165. PMID: 30637916
- Analyses of Antioxidative Properties of Selected Cyclitols and Their Mixtures with Flavanones and Glutathione. | Płonka, J., et al. 2021. Molecules. 27: PMID: 35011390
- Synthesis of 1L-chiro-inositol 2,3,5-trisphosphorothioate, the first partial agonist at the platelet myo-inositol 1,4,5-trisphosphate receptor. | Liu, C., et al. 1994. Bioorg Med Chem. 2: 253-7. PMID: 7922136
Activator of:
CRIP3.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
L-Quebrachitol, 1 g | sc-205950 | 1 g | $151.00 |
