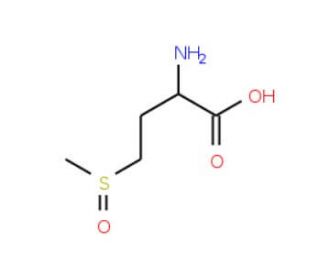
L-Methionine sulfoxide (CAS 3226-65-1)
QUICK LINKS
L-Methionine sulfoxide is a compound that functions as an antioxidant in experimental applications. It acts by scavenging free radicals and reactive oxygen species, thereby protecting cells and biomolecules from oxidative damage. L-Methionine Sulfoxide′s mechanism of action involves the reduction of hydrogen peroxide and other reactive oxygen species, preventing them from causing cellular damage. L-Methionine sulfoxide achieves this by undergoing a redox reaction, where it is converted back to its reduced form, L-Methionine, after neutralizing the reactive oxygen species. L-Methionine Sulfoxide′s functional role lies in its ability to serve as a model molecule for studying antioxidant mechanisms and oxidative stress in biological systems. Its mechanism of action at the molecular level involves the direct interaction with reactive oxygen species, leading to the restoration of cellular redox balance.
L-Methionine sulfoxide (CAS 3226-65-1) References
- Identification and characterization of a putative active site for peptide methionine sulfoxide reductase (MsrA) and its substrate stereospecificity. | Moskovitz, J., et al. 2000. J Biol Chem. 275: 14167-72. PMID: 10799493
- Stereospecific micellar electrokinetic chromatography assay of methionine sulfoxide reductase activity employing a multiple layer coated capillary. | Zhu, Q., et al. 2013. Electrophoresis. 34: 2712-7. PMID: 23775584
- Stereospecific electrophoretically mediated microanalysis assay for methionine sulfoxide reductase enzymes. | Zhu, Q., et al. 2014. Anal Bioanal Chem. 406: 1723-9. PMID: 24424966
- Impurity profiling of L-methionine by HPLC on a mixed mode column. | Kühnreich, R. and Holzgrabe, U. 2016. J Pharm Biomed Anal. 122: 118-25. PMID: 26852161
- Oryzamides A-E, Cyclodepsipeptides from the Sponge-Derived Fungus Nigrospora oryzae PF18. | Ding, LJ., et al. 2016. J Nat Prod. 79: 2045-52. PMID: 27489998
- Nutritional value and safety of methionine derivatives, isomeric dipeptides and hydroxy analogs in mice. | Friedman, M. and Gumbmann, MR. 1988. J Nutr. 118: 388-97. PMID: 3351635
- The Function of Selenium in Central Nervous System: Lessons from MsrB1 Knockout Mouse Models. | Shi, T., et al. 2021. Molecules. 26: PMID: 33806413
- A biocompatible cell cryoprotectant based on sulfoxide-containing amino acids: mechanism and application. | Liu, M., et al. 2023. J Mater Chem B. 11: 2504-2517. PMID: 36852742
- Utilization of L-methionine sulfoxide by the rat. | Miller, SA., et al. 1970. J Nutr. 100: 909-15. PMID: 5495843
- The reduction of S-methyl-L-cysteine sulfoxide and L-methionine sulfoxide in turnip and bean leaves. | Doney, RC. and Thompson, JF. 1966. Biochim Biophys Acta. 124: 39-49. PMID: 5966721
- Availability of oxidized sulfur amino acids for the growing chick. | Kuzmicky, DD., et al. 1977. Poult Sci. 56: 1560-5. PMID: 605099
- Poly-L-methionine sulfoxide: a biologically inert analogue of dimethyl sulfoxide with solubilizing potency. | Pitha, J., et al. 1983. J Pharm Sci. 72: 665-8. PMID: 6576129
- Utilization of L-methionine sulfoxide, L-methionine sulfone and cysteic acid by the weanling rat. | Anderson, GH., et al. 1976. J Nutr. 106: 1108-14. PMID: 939991
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
L-Methionine sulfoxide, 5 g | sc-300889 | 5 g | $88.00 | |||
L-Methionine sulfoxide, 25 g | sc-300889A | 25 g | $352.00 |
