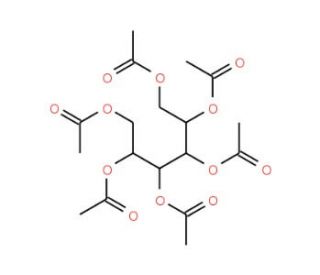
L-Iditol Hexaacetate
QUICK LINKS
L-Iditol Hexaacetate, also known as D-Sorbitol Hexaacetate, is a chemical compound commonly utilized in organic synthesis and carbohydrate chemistry research. Its primary application lies in the protection of hydroxyl groups in carbohydrates, particularly in the context of carbohydrate derivatization and modification. The hexaacetylation of L-iditol involves the replacement of all six hydroxyl groups with acetate moieties, rendering the resulting compound more stable and less reactive towards other chemical reactions. This chemical modification serves various purposes in research, including facilitating the isolation and purification of specific carbohydrate derivatives, enhancing the solubility and chromatographic properties of carbohydrate compounds, and enabling the selective modification of hydroxyl groups for further synthetic transformations. L-Iditol Hexaacetate has found extensive use in the synthesis of complex carbohydrate structures, glycosylation reactions, and the preparation of carbohydrate-based materials for applications in fields such as drug delivery, biomaterials science, and carbohydrate-based nanotechnology. Furthermore, its role as a protecting group in carbohydrate chemistry enables the synthesis of structurally diverse carbohydrate derivatives with tailored properties and functionalities, contributing to advancements in areas such as glycobiology, chemical biology, and materials science.
L-Iditol Hexaacetate References
- A definitive synthesis of D-myo-inositol 1,4,5,6-tetrakisphosphate and its enantiomer D-myo-inositol 3,4,5,6-tetrakisphosphate from a novel butane-2,3-diacetal-protected inositol. | Mills, SJ., et al. 2003. Chemistry. 9: 6207-14. PMID: 14679531
- Synthesis of L-idaro-1,4-lactone, an inhibitor of alpha-L-idosiduronase. | Herd, JK., et al. 1982. Carbohydr Res. 99: 33-9. PMID: 7055824
- The Structure and Configuration of Resin Acids. Podocarpic Acid and Ferruginol | William P. Campbell and David Todd. 1942. Journal of the American Chemical Society. 64: 928–935.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
L-Iditol Hexaacetate, 200 mg | sc-280884 | 200 mg | $300.00 |
