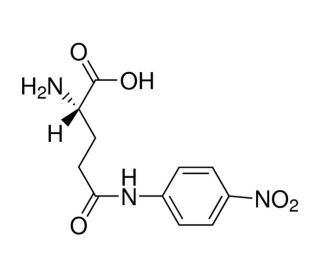
L-Glutamic acid γ-(4-nitroanilide) (CAS 7300-59-6)
QUICK LINKS
L-Glutamic acid γ-(4-nitroanilide) is a compound that functions as a substrate for the enzyme γ-glutamyltransferase (GGT) in various biochemical assays. When used in experimental applications, L-Glutamic acid γ-(4-nitroanilide) serves as a specific substrate for GGT, allowing for the measurement of GGT activity in biological samples. Upon interaction with GGT, L-Glutamic acid γ-(4-nitroanilide) undergoes enzymatic cleavage, resulting in the release of 4-nitroaniline. This reaction can be quantitatively measured using spectrophotometric methods, providing insights into GGT activity levels in biological systems. L-Glutamic acid γ-(4-nitroanilide)′s mechanism of action involves its specific recognition and cleavage by the GGT enzyme, leading to the generation of a measurable product.
L-Glutamic acid γ-(4-nitroanilide) (CAS 7300-59-6) References
- Nonlinear free energy relationship in the general-acid-catalyzed acylation of rat kidney gamma-glutamyl transpeptidase by a series of gamma-glutamyl anilide substrate analogues. | Ménard, A., et al. 2001. Biochemistry. 40: 12678-85. PMID: 11601992
- Characterization of Helicobacter pylori gamma-glutamyltranspeptidase reveals the molecular basis for substrate specificity and a critical role for the tyrosine 433-containing loop in catalysis. | Morrow, AL., et al. 2007. Biochemistry. 46: 13407-14. PMID: 17960917
- Crystal structure of acivicin-inhibited gamma-glutamyltranspeptidase reveals critical roles for its C-terminus in autoprocessing and catalysis. | Williams, K., et al. 2009. Biochemistry. 48: 2459-67. PMID: 19256527
- Biochemical and structural properties of gamma-glutamyl transpeptidase from Geobacillus thermodenitrificans: an enzyme specialized in hydrolase activity. | Castellano, I., et al. 2010. Biochimie. 92: 464-74. PMID: 20138205
- Gene cloning and protein expression of γ-glutamyltranspeptidases from Thermus thermophilus and Deinococcus radiodurans: comparison of molecular and structural properties with mesophilic counterparts. | Castellano, I., et al. 2011. Extremophiles. 15: 259-70. PMID: 21298394
- Characterization of MtfA, a novel regulatory output signal protein of the glucose-phosphotransferase system in Escherichia coli K-12. | Göhler, AK., et al. 2012. J Bacteriol. 194: 1024-35. PMID: 22178967
- γ-Glutamyltranspeptidases: sequence, structure, biochemical properties, and biotechnological applications. | Castellano, I. and Merlino, A. 2012. Cell Mol Life Sci. 69: 3381-94. PMID: 22527720
- Donor substrate specificity of bovine kidney gamma-glutamyltransferase. | Agblor, AA. and Josephy, PD. 2013. Chem Biol Interact. 203: 480-5. PMID: 23500672
- Partial purification and properties of gamma-glutamyltranspeptidase from mycelia of Morchella esculenta. | Moriguchi, M., et al. 1986. Arch Microbiol. 144: 15-9. PMID: 2870692
- Behaviour of L-gamma-glutamyl-4-nitroanilide and L-gamma-glutamyl-3-carboxy-4-nitroanilide with respect to gamma-glutamyltransferases of different origin. | Verhoeff, PM., et al. 1988. Clin Chim Acta. 175: 129-34. PMID: 2900705
- The distribution and function characterization of the i type lysozyme from Apostichopus japonicus. | Li, C., et al. 2018. Fish Shellfish Immunol. 74: 419-425. PMID: 29079205
- A Photo-Crosslinkable Kidney ECM-Derived Bioink Accelerates Renal Tissue Formation. | Ali, M., et al. 2019. Adv Healthc Mater. 8: e1800992. PMID: 30725520
- Copper biodistribution after acute systemic administration of copper gluconate to rats. | García-Martínez, BA., et al. 2021. Biometals. 34: 687-700. PMID: 33900531
- Tridegin, a new peptidic inhibitor of factor XIIIa, from the blood-sucking leech Haementeria ghilianii. | Finney, S., et al. 1997. Biochem J. 324 (Pt 3): 797-805. PMID: 9210403
- Analysis of kinetic properties of gamma-glutamyl transpeptidase from rat kidney. | Dvoráková, L., et al. 1996. Gen Physiol Biophys. 15: 403-13. PMID: 9228521
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
L-Glutamic acid γ-(4-nitroanilide), 1 g | sc-250219 | 1 g | $48.00 |
