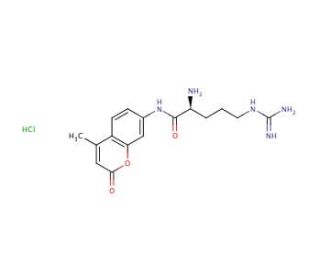
L-Arginine-7-amido-4-methylcoumarin hydrochloride (CAS 69304-16-1)
QUICK LINKS
L-Arginine-7-amido-4-methylcoumarin hydrochloride (LAMCH) is a synthetic compound used in scientific research as a substrate of cathepsin H. Derived from the naturally occurring amino acid L-arginine, L-Arginine-7-amido-4-methylcoumarin hydrochloride has found applications in various laboratory experiments involving biochemistry, physiology, and pharmacology. In scientific research, L-Arginine-7-amido-4-methylcoumarin hydrochloride has been utilized in multiple contexts. As a fluorescent probe, it enables the examination of protein structure and function, as well as the investigation of protein interactions with other molecules. It serves as useful for studying ligand binding to receptors and exploring the kinetics of enzyme-catalyzed reactions. Moreover, L-Arginine-7-amido-4-methylcoumarin hydrochloride facilitates the examination of drug effects on diverse biochemical pathways. Although the precise mechanism of action of L-Arginine-7-amido-4-methylcoumarin hydrochloride remains incompletely understood, it is believed to function by binding to specific receptors on cell surfaces, initiating a signaling cascade that leads to various physiological and biochemical effects. The specific effects of L-Arginine-7-amido-4-methylcoumarin hydrochloride depend on the nature of the experiment being conducted. Generally, it has demonstrated influence over numerous biochemical pathways involved in cell signaling, gene expression, metabolism, and cell proliferation. Additionally, it may act to impact physiological processes such as blood pressure, heart rate, and respiration.
L-Arginine-7-amido-4-methylcoumarin hydrochloride (CAS 69304-16-1) References
- Purification and characterization of an arginine aminopeptidase from Lactobacillus sakei. | Sanz, Y. and Toldrá, F. 2002. Appl Environ Microbiol. 68: 1980-7. PMID: 11916721
- Overexpression of peptidases in Lactococcus and evaluation of their release from leaky cells. | Tuler, TR., et al. 2002. J Dairy Sci. 85: 2438-50. PMID: 12416795
- Antibacterial and proteolytic activity in venom from the endoparasitic wasp Pimpla hypochondriaca (Hymenoptera: Ichneumonidae). | Dani, MP., et al. 2003. J Insect Physiol. 49: 945-54. PMID: 14511827
- Purification and functional characterisation of rhiminopeptidase A, a novel aminopeptidase from the venom of Bitis gabonica rhinoceros. | Vaiyapuri, S., et al. 2010. PLoS Negl Trop Dis. 4: e796. PMID: 20706583
- Physical exercise and a leucine-rich diet modulate the muscle protein metabolism in Walker tumor-bearing rats. | Salomão, EM., et al. 2010. Nutr Cancer. 62: 1095-104. PMID: 21058197
- Effect of selection for growth rate on the ageing of myofibrils, meat texture properties and the muscle proteolytic potential of m. longissimus in rabbits. | Gil, M., et al. 2006. Meat Sci. 72: 121-9. PMID: 22061382
- Dietary tools to modulate glycogen storage in gilthead seabream muscle: glycerol supplementation. | Silva, TS., et al. 2012. J Agric Food Chem. 60: 10613-24. PMID: 22994592
- Proteolytic activities in fillets of selected underutilized Australian fish species. | Ahmed, Z., et al. 2013. Food Chem. 140: 238-44. PMID: 23578639
- Changes in Cathepsin Activity during Low-Temperature Storage and Sous Vide Processing of Beef Brisket. | Kaur, L., et al. 2020. Food Sci Anim Resour. 40: 415-425. PMID: 32426720
- Separation of a new alpha-N-benzoylarginine-beta-naphthylamide hydrolase from cathepsin B1. Purification, characterization, and properties of both enzymes from rabbit lung. | Singh, H. and Kalnitsky, G. 1978. J Biol Chem. 253: 4319-26. PMID: 659418
- Fluorimetric assays for cathepsin B and cathepsin H with methylcoumarylamide substrates. | Barrett, AJ. 1980. Biochem J. 187: 909-12. PMID: 6897924
- Cathepsin B, Cathepsin H, and cathepsin L. | Barrett, AJ. and Kirschke, H. 1981. Methods Enzymol. 80 Pt C: 535-61. PMID: 7043200
- Impairment of multicycle influenza virus growth in Vero (WHO) cells by loss of trypsin activity. | Kaverin, NV. and Webster, RG. 1995. J Virol. 69: 2700-3. PMID: 7884927
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
L-Arginine-7-amido-4-methylcoumarin hydrochloride, 5 mg | sc-215211 | 5 mg | $107.00 | |||
L-Arginine-7-amido-4-methylcoumarin hydrochloride, 25 mg | sc-215211A | 25 mg | $233.00 |
