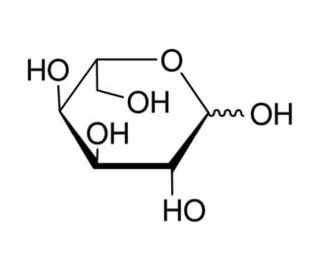
L-Altrose (CAS 1949-88-8)
QUICK LINKS
L-Altrose, a rare naturally occurring aldohexose, plays a significant role in the research fields of carbohydrate chemistry and glycobiology. As one of the lesser-studied sugars due to its rarity and instability in its pyranose form, L-altrose offers unique opportunities for studying stereochemical variation and its impact on biological interactions. In research, L-altrose is primarily utilized in the synthesis of oligosaccharides and glycoconjugates, providing insights into how slight variations in sugar configuration influence molecular recognition processes. Scientists employ this monosaccharide to explore the enzymatic pathways of sugar metabolism, as its unusual stereochemistry can influence enzyme specificity and reaction pathways, thereby broadening our understanding of carbohydrate active enzymes. Additionally, L-altrose is used in the design of mimetics that can interfere with or modulate biological processes involving sugars, such as bacterial adhesion to host cells, which is mediated through carbohydrate-lectin interactions. This research provides valuable data on the structural requirements for biological activity and opens up potential applications in developing novel biochemical tools that could influence glycan-mediated biological functions. Thus, L-altrose is integral in advancing glycobiological research and deepening our comprehension of carbohydrate-mediated biological phenomena.
L-Altrose (CAS 1949-88-8) References
- Integrated route to the L-aldohexoses using a common man-made chiral building block. | Takeuchi, M., et al. 2000. Chirality. 12: 338-41. PMID: 10824148
- L-Altruronic acid formed by epimerization of D-galacturonic acid methyl esters during saponification of citrus pectin. | Zhan, D., et al. 2001. Carbohydr Res. 330: 357-63. PMID: 11270814
- Creation of the first anomeric D/L-sugar kinase by means of directed evolution. | Hoffmeister, D., et al. 2003. Proc Natl Acad Sci U S A. 100: 13184-9. PMID: 14612558
- A versatile route to L-hexoses: synthesis of L-mannose and L-altrose. | Guaragna, A., et al. 2006. Org Lett. 8: 4863-6. PMID: 17020322
- The altrose group of substances. | RICHTMYER, NK. 1945. Adv Carbohydr Chem. 1: 37-76. PMID: 21020773
- Characterization of a recombinant cellobiose 2-epimerase from Caldicellulosiruptor saccharolyticus and its application in the production of mannose from glucose. | Park, CS., et al. 2011. Appl Microbiol Biotechnol. 92: 1187-96. PMID: 21691788
- Synthesis of 5-C-Methylated d-Mannose, d-Galactose, l-Gulose, and l-Altrose and Their Structural Elucidation by NMR Spectroscopy. | Köllmann, C., et al. 2018. Org Lett. 20: 1220-1223. PMID: 29406726
- Neutral sugar composition of extracellular polysaccharides produced by strains of Butyrivibrio fibrisolvens. | Stack, RJ. 1988. Appl Environ Microbiol. 54: 878-83. PMID: 3377501
- Synthesis of L‐Altrose and Some Derivatives. | Lunau, Nathalie and Chris Meier. 2012. European Journal of Organic Chemistry. 2012.31: 6260-6270.
- Three-step synthesis of protected L-altrose from D-galactose derived Perlin aldehyde. | Halder, Suvendu, et al. 2020. J. Indian Chem. Soc. 97: 243-250.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
L-Altrose, 25 mg | sc-257648 | 25 mg | $112.00 |
