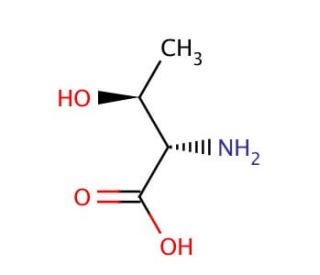
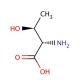
L-allo-Threonine (CAS 28954-12-3)
QUICK LINKS
L-allo-Threonine is an isomer of the amino acid threonine, distinguished by the arrangement of its molecular components, which imparts unique properties that have become a focal point in biochemical and structural biology research. Unlike its counterpart, L-threonine, which is incorporated into proteins during translation, L-allo-threonine is not found in proteins and thus serves as a tool for probing protein structure, function, and stability. Researchers leverage L-allo-Threonine in the study of enzyme specificity, exploring how enzymes distinguish between closely related substrates. This isomer also plays a critical role in synthetic biology, where it is used to engineer novel proteins with altered amino acid sequences, enabling the investigation of protein folding mechanisms and the design of proteins with new functions. Additionally, L-allo-Threonine′s unique structural properties make it a valuable component in the development of peptidomimetics, molecules that mimic the structure of peptides and are used to elucidate interactions at the molecular level, offering insights into the fundamental principles of molecular recognition and binding.
L-allo-Threonine (CAS 28954-12-3) References
- X-ray structures of threonine aldolase complexes: structural basis of substrate recognition. | Kielkopf, CL. and Burley, SK. 2002. Biochemistry. 41: 11711-20. PMID: 12269813
- Characterization of short-chain dehydrogenase/reductase homologues of Escherichia coli (YdfG) and Saccharomyces cerevisiae (YMR226C). | Fujisawa, H., et al. 2003. Biochim Biophys Acta. 1645: 89-94. PMID: 12535615
- Towards preparative asymmetric synthesis of beta-hydroxy-alpha-amino acids: L-allo-threonine formation from glycine and acetaldehyde using recombinant GlyA. | Makart, S., et al. 2007. J Biotechnol. 130: 402-10. PMID: 17597243
- Assessment of threonine metabolism in vivo by gas chromatography/mass spectrometry and stable isotope infusion. | Ballèvre, O., et al. 1991. Anal Biochem. 193: 212-9. PMID: 1908193
- Discovery and characterization of D-phenylserine deaminase from Arthrobacter sp. TKS1. | Muramatsu, H., et al. 2011. Appl Microbiol Biotechnol. 90: 159-72. PMID: 21190106
- Alleviation of asthma-related symptoms by a derivative of L-allo threonine. | Heo, JC. and Lee, SH. 2013. Int J Mol Med. 31: 881-7. PMID: 23381610
- Arg-265: a critical residue of L.donovani cytosolic SHMT in maintaining the binding of THF and catalysis. | Gandhi, S., et al. 2015. Exp Parasitol. 149: 16-23. PMID: 25499510
- The Important Role of the Hydroxyl Group on the Conformational Adaptability in Bis(l-threoninato)copper(II) Compared to Bis(l-allo-threoninato)copper(II): Quantum Chemical Study. | Marković, M., et al. 2016. Inorg Chem. 55: 7694-708. PMID: 27442350
- Effect of Novel Compound LX519290, a Derivative of l-allo Threonine, on Antioxidant Potential in Vitro and in Vivo. | Chun, K., et al. 2016. Int J Mol Sci. 17: PMID: 27598126
- L-Threonine and its analogue added to autoclaved solid medium suppress trichothecene production by Fusarium graminearum. | Maeda, K., et al. 2017. Arch Microbiol. 199: 945-952. PMID: 28357472
- Biosynthesis of Cyclochlorotine: Identification of the Genes Involved in Oxidative Transformations and Intramolecular O,N-Transacylation. | Jiang, Y., et al. 2021. Org Lett. 23: 2616-2620. PMID: 33736433
- Terahertz Spectroscopy Study of the Stereoisomers of Threonine. | Zeng, R., et al. 2022. Appl Spectrosc. 76: 1132-1141. PMID: 35469427
- Interactions between copper (II) ions and L-threonine, L-allo-threonine and L-serine in aqueous solution. | Grenouillet, P., et al. 1973. Biochim Biophys Acta. 322: 185-94. PMID: 4765086
- Globomycin, a new peptide antibiotic with spheroplast-forming activity. III. Structural determination of globomycin. | Nakajima, M., et al. 1978. J Antibiot (Tokyo). 31: 426-32. PMID: 670085
- Gene cloning, biochemical characterization and physiological role of a thermostable low-specificity L-threonine aldolase from Escherichia coli. | Liu, JQ., et al. 1998. Eur J Biochem. 255: 220-6. PMID: 9692922
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
L-allo-Threonine, 250 mg | sc-255235 | 250 mg | $281.00 |
