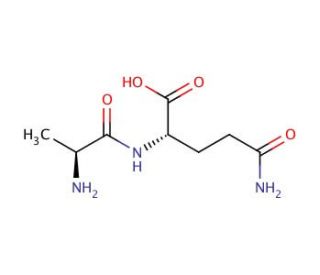
L-alanyl-L-glutamine (CAS 39537-23-0)
QUICK LINKS
L-alanyl-L-glutamine is a dipeptide consisting of the amino acids alanine and glutamine. This compound is of particular interest in scientific research due to its enhanced stability compared to L-glutamine alone, making it a preferred alternative in various experimental settings. In aqueous solutions, L-glutamine is relatively unstable and can degrade to form ammonia and pyroglutamic acid; however, when bonded with alanine to form L-alanyl-L-glutamine, the stability is significantly improved, thereby reducing these degradation issues. The stability of L-alanyl-L-glutamine has facilitated its use in studies that require a controlled environment for cell cultures. It is used extensively in in vitro experiments to understand cellular metabolism and physiology, particularly under conditions that demand sustained availability of glutamine, which is critical for rapidly dividing cells such as immune and intestinal cells. In scientific research, L-alanyl-L-glutamine serves not just as a nutrient that supports cellular growth and function, but also as a tool to study the metabolism of amino acids in various cell types. This dipeptide is especially useful in studying the role of glutamine as a cellular fuel and its importance in protein synthesis and energy production. Moreover, it aids in the investigation of the transport mechanisms of peptides and their metabolic effects, providing insights into how cells regulate nutrient uptake and utilization.
L-alanyl-L-glutamine (CAS 39537-23-0) References
- Quantification of l-alanyl-l-glutamine in mammalian cell culture broth: Evaluation of different detectors. | Krömer, JO., et al. 2011. Anal Biochem. 416: 129-31. PMID: 21651886
- Preconditioning with L-alanyl-L-glutamine in a Mongolian gerbil model of acute cerebral ischemia/reperfusion injury. | Pires, VL., et al. 2011. Acta Cir Bras. 26 Suppl 1: 14-20. PMID: 21971651
- Enzymatic production of L-alanyl-L-glutamine by recombinant E. coli expressing α-amino acid ester acyltransferase from Sphingobacterium siyangensis. | Hirao, Y., et al. 2013. Biosci Biotechnol Biochem. 77: 618-23. PMID: 23470770
- Oral supplementations with L-glutamine or L-alanyl-L-glutamine do not change metabolic alterations induced by long-term high-fat diet in the B6.129F2/J mouse model of insulin resistance. | Bock, PM., et al. 2016. Mol Cell Biochem. 411: 351-62. PMID: 26530165
- Evitar (l-Alanyl-l-Glutamine) Regulates Key Signaling Molecules in the Pathogenesis of Postoperative Tissue Fibrosis. | Robertson, LM., et al. 2019. Reprod Sci. 26: 724-733. PMID: 30185141
- Metabolic engineering of Escherichia coli for efficient production of L-alanyl-L-glutamine. | Zhu, J., et al. 2020. Microb Cell Fact. 19: 129. PMID: 32527330
- Production of L-alanyl-L-glutamine by immobilized Escherichia coli expressing amino acid ester acyltransferase. | Pei, X., et al. 2020. Appl Microbiol Biotechnol. 104: 6967-6976. PMID: 32594215
- Clean Production of l-Alanyl-l-glutamine by an Efficient Yeast Biocatalyst Expressing α-Amino Acid Ester Acyltransferase without N-Glycosylation. | Li, Y., et al. 2023. J Agric Food Chem. 71: 6398-6405. PMID: 37027821
- Cerium ions immobilized magnetic graphite nitride decorated with L-Alanyl-L-Glutamine as new chelator for enrichment of phosphopeptides. | Jiang, D., et al. 2023. Mikrochim Acta. 190: 452. PMID: 37882891
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
L-alanyl-L-glutamine, 1 g | sc-337885 | 1 g | $772.00 |
