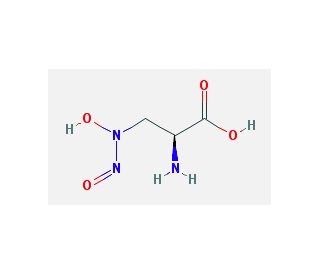
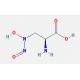
L-Alanosine (CAS 5854-93-3)
See product citations (2)
QUICK LINKS
L-Alanosine is an amino acid analogue with notable interest in molecular biology research for its role as an antimetabolite that disrupts nucleotide biosynthesis. By inhibiting adenylosuccinate synthetase, an enzyme for the de novo synthesis of purine nucleotides, L-Alanosine serves as a tool for dissecting the purine biosynthetic pathway. Its utility extends to the study of cellular resistance mechanisms against antimetabolites, helping to elucidate how cells modify metabolic pathways in response to metabolic stress. In addition, this compound aids in the investigation of fundamental biological processes such as DNA replication and RNA transcription by affecting the availability of purine nucleotides. L-Alanosine also contributes to the field of synthetic biology, where it is used in constructing novel metabolic pathways and synthesizing purine nucleotide analogues to explore or influence enzyme activity.
L-Alanosine (CAS 5854-93-3) References
- Determination of derivatized l-alanosine in plasma by liquid chromatography-tandem mass spectrometry. | Gantverg, A., et al. 2004. J Chromatogr B Analyt Technol Biomed Life Sci. 803: 311-5. PMID: 15063341
- Status of methylthioadenosine phosphorylase and its impact on cellular response to L-alanosine and methylmercaptopurine riboside in human soft tissue sarcoma cells. | Li, W., et al. 2004. Oncol Res. 14: 373-9. PMID: 15301428
- EFA (9-beta-D-erythrofuranosyladenine) is an effective salvage agent for methylthioadenosine phosphorylase-selective therapy of T-cell acute lymphoblastic leukemia with L-alanosine. | Batova, A., et al. 2006. Blood. 107: 898-903. PMID: 16234352
- Circadian pharmacology of L-alanosine (SDX-102) in mice. | Li, XM., et al. 2006. Mol Cancer Ther. 5: 337-46. PMID: 16505107
- Increased tumor control rates in murine fibrosarcoma by combined therapy with L-alanosine and radiation. | Hong, SS., et al. 1989. Jpn J Cancer Res. 80: 592-6. PMID: 2503478
- The l-Alanosine Gene Cluster Encodes a Pathway for Diazeniumdiolate Biosynthesis. | Ng, TL., et al. 2020. Chembiochem. 21: 1155-1160. PMID: 31643127
- Biosynthesis of the N-N-Bond-Containing Compound l-Alanosine. | Wang, M., et al. 2020. Angew Chem Int Ed Engl. 59: 3881-3885. PMID: 31823464
- Purine Synthesis Inhibitor L-Alanosine Impairs Mitochondrial Function and Stemness of Brain Tumor Initiating Cells. | Singh, SX., et al. 2022. Biomedicines. 10: PMID: 35453502
- L-alanosine: a noncooperative substrate for Escherichia coli aspartate transcarbamylase. | Baillon, J., et al. 1985. Biochemistry. 24: 7182-7. PMID: 3910096
- Effect of L-alanosine on immune response. | Mistrello, G. and Bassi, L. 1984. J Immunopharmacol. 6: 25-41. PMID: 6470495
- Antitumor effect of L-alanosine (NSC 153553) on sensitive and resistant sublines of murine leukemias. | Chitnis, MP., et al. 1984. Tumori. 70: 317-20. PMID: 6474581
- Synthesis and in vitro biological evaluation of N-[(5-amino-1-beta-D-ribofuranosyl-1H-imidazol-4-yl)carbonyl]-3- (hydroxynitrosamino)-L-alanine (L-alanosine AICO ribonucleoside). | Strazzolini, P., et al. 1984. J Med Chem. 27: 1295-9. PMID: 6592359
- Modulation of fluoropyrimidine metabolism in L1210 cells by L-alanosine. | Heimer, R., et al. 1983. Biochem Pharmacol. 32: 199-206. PMID: 6870949
- Determinants of the toxicity of L-alanosine to various organs of the mouse. | Tyagi, AK., et al. 1981. Toxicology. 21: 59-69. PMID: 7281196
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
L-Alanosine, 5 mg | sc-207791 | 5 mg | $377.00 |
