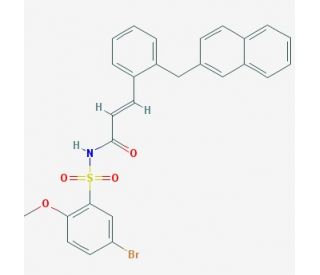
L-798,106 (CAS 244101-02-8)
See product citations (16)
QUICK LINKS
L-798,106 is a chemical used primarily in research focused on the endocrine system, where it acts as a selective antagonist for the cholecystokinin receptor subtype known as CCK2. By blocking this receptor, L-798,106 helps scientists to study the physiological roles of cholecystokinin, particularly in the context of digestion and satiety. The compound is instrumental in investigating the regulation of gastric acid secretion and the motility of the gastrointestinal tract. In addition, L-798,106 is utilized in research aiming to understand the neuromodulatory functions of cholecystokinin in the brain, including its potential to influence anxiety and pain perception. Through the use of L-798,106, researchers can dissect the complex signaling pathways mediated by the CCK2 receptor and its involvement in various biological processes.
L-798,106 (CAS 244101-02-8) References
- Structure-activity relationship of cinnamic acylsulfonamide analogues on the human EP3 prostanoid receptor. | Juteau, H., et al. 2001. Bioorg Med Chem. 9: 1977-84. PMID: 11504634
- E-ring 8-isoprostanes inhibit ACh release from parasympathetic nerves innervating guinea-pig trachea through agonism of prostanoid receptors of the EP3-subtype. | Clarke, DL., et al. 2004. Br J Pharmacol. 141: 600-9. PMID: 14744812
- Activation of prostaglandin EP receptors by lubiprostone in rat and human stomach and colon. | Bassil, AK., et al. 2008. Br J Pharmacol. 154: 126-35. PMID: 18332851
- Possible involvement of brain prostaglandin E2 and prostanoid EP3 receptors in prostaglandin E2 glycerol ester-induced activation of central sympathetic outflow in the rat. | Shimizu, T., et al. 2014. Neuropharmacology. 82: 19-27. PMID: 24657150
- Prostaglandin E2 receptor 3 signaling is induced in placentas with unexplained recurrent pregnancy losses. | Ye, Y., et al. 2018. Endocr Connect. 7: 749-761. PMID: 29700097
- The role of Cox-2 and prostaglandin E2 receptor EP3 in pancreatic β-cell death. | Amior, L., et al. 2019. FASEB J. 33: 4975-4986. PMID: 30629897
- Generation of Cellular Reactive Oxygen Species by Activation of the EP2 Receptor Contributes to Prostaglandin E2-Induced Cytotoxicity in Motor Neuron-Like NSC-34 Cells. | Kosuge, Y., et al. 2020. Oxid Med Cell Longev. 2020: 6101838. PMID: 32411331
- The PGE2 receptor EP3 plays a positive role in the activation of hypothalamic-pituitary-adrenal axis and neuronal activity in the hypothalamus under immobilization stress. | Lv, L., et al. 2021. Brain Res Bull. 168: 45-51. PMID: 33370588
- Prostaglandin E2 sensitizes the cough reflex centrally via EP3 receptor-dependent activation of NaV 1.8 channels. | Al-Kandery, AA., et al. 2021. Respir Res. 22: 296. PMID: 34794450
- Docosahexaenoic acid and eicosapentaenoic acid strongly inhibit prostanoid TP receptor-dependent contractions of guinea pig gastric fundus smooth muscle. | Xu, K., et al. 2022. Pharmacol Res Perspect. 10: e00952. PMID: 35466586
- The EP3 and EP4 Receptor Subtypes both Mediate the Fever-producing Effects of Prostaglandin E2 in the Rostral Ventromedial Preoptic Area of the Hypothalamus in Rats. | Osaka, T. 2022. Neuroscience. 494: 25-37. PMID: 35550162
- Effects of NP-1815-PX, a P2X4 Receptor Antagonist, on Contractions in Guinea Pig Tracheal and Bronchial Smooth Muscles. | Obara, K., et al. 2022. Biol Pharm Bull. 45: 1158-1165. PMID: 35908896
- Inhibition of cyclooxygenase and EP3 receptor improved long term potentiation in a rat organotypic hippocampal model of repeated blast traumatic brain injury. | Varghese, N. and Morrison, B. 2023. Neurochem Int. 163: 105472. PMID: 36599378
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
L-798,106, 5 mg | sc-204047 | 5 mg | $105.00 | |||
L-798,106, 25 mg | sc-204047A | 25 mg | $417.00 |
