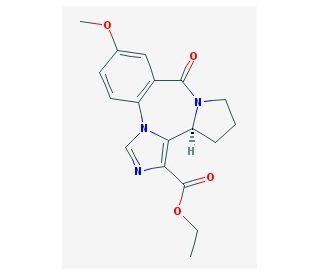
Molecular structure of L-655,708, CAS Number: 130477-52-0
L-655,708 (CAS 130477-52-0)
Alternate Names:
Ethyl (S)-11,12,13,13a-Tetrahydro-7-methoxy-9-oxo-9H-imidazo[1,5-a]pyrrolo[2,1-c][1,4]benzodiazepine-1-carboxylate; L-655708
Application:
L-655,708 is a potent and selective inverse agonist for the benzodiazepine
CAS Number:
130477-52-0
Purity:
≥98%
Molecular Weight:
341.37
Molecular Formula:
C18H19N3O4
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
L-655,708 is a synthetically crafted molecule that is known to interact with specific receptors, such as G-protein coupled receptors (GPCRs). Its role in the scientific study extends to the evaluation of molecular responses and receptor-mediated signaling, which are critical in the field of biochemistry and molecular biology. The interaction with these receptors triggers a series of biochemical events that lead to observable outcomes in studies.
L-655,708 (CAS 130477-52-0) References
- Identification of amino acid residues responsible for the alpha5 subunit binding selectivity of L-655,708, a benzodiazepine binding site ligand at the GABA(A) receptor. | Casula, MA., et al. 2001. J Neurochem. 77: 445-51. PMID: 11299307
- Anxiogenic-like activity of L-655,708, a selective ligand for the benzodiazepine site of GABA(A) receptors which contain the alpha-5 subunit, in the elevated plus-maze test. | Navarro, JF., et al. 2002. Prog Neuropsychopharmacol Biol Psychiatry. 26: 1389-92. PMID: 12502028
- Rat pharmacokinetics and pharmacodynamics of a sustained release formulation of the GABAA alpha5-selective compound L-655,708. | Atack, JR., et al. 2006. Drug Metab Dispos. 34: 887-93. PMID: 16455808
- L-655,708 enhances cognition in rats but is not proconvulsant at a dose selective for alpha5-containing GABAA receptors. | Atack, JR., et al. 2006. Neuropharmacology. 51: 1023-9. PMID: 17046030
- Short-term memory impairment after isoflurane in mice is prevented by the α5 γ-aminobutyric acid type A receptor inverse agonist L-655,708. | Saab, BJ., et al. 2010. Anesthesiology. 113: 1061-71. PMID: 20966663
- Potentiation of GABAA receptor activity by volatile anaesthetics is reduced by α5GABAA receptor-preferring inverse agonists. | Lecker, I., et al. 2013. Br J Anaesth. 110 Suppl 1: i73-81. PMID: 23535829
- Selective Pharmacological Augmentation of Hippocampal Activity Produces a Sustained Antidepressant-Like Response without Abuse-Related or Psychotomimetic Effects. | Carreno, FR., et al. 2017. Int J Neuropsychopharmacol. 20: 504-509. PMID: 28339593
- Inhibition of α5 GABAA receptors has preventive but not therapeutic effects on isoflurane-induced memory impairment in aged rats. | Zhao, ZF., et al. 2019. Neural Regen Res. 14: 1029-1036. PMID: 30762015
- Contribution of GABAA receptor subunits to attention and social behavior. | Paine, TA., et al. 2020. Behav Brain Res. 378: 112261. PMID: 31560920
- Mechanisms associated with the antidepressant-like effects of L-655,708. | Bugay, V., et al. 2020. Neuropsychopharmacology. 45: 2289-2298. PMID: 32688367
- A multi-dosing regimen to enhance the spatial memory of normal rats with α5-containing GABAA receptor negative allosteric modulator L-655,708. | Yuan, C., et al. 2021. Psychopharmacology (Berl). 238: 3375-3389. PMID: 34389882
- Modulation of relapse-like drinking in male Sprague-Dawley rats by ligands targeting the α5GABAA receptor. | Chandler, CM., et al. 2021. Neuropharmacology. 199: 108785. PMID: 34509495
- MD2 contributes to the pathogenesis of perioperative neurocognitive disorder via the regulation of α5GABAA receptors in aged mice. | Zuo, W., et al. 2021. J Neuroinflammation. 18: 204. PMID: 34530841
- [3H]L-655,708, a novel ligand selective for the benzodiazepine site of GABAA receptors which contain the alpha 5 subunit. | Quirk, K., et al. 1996. Neuropharmacology. 35: 1331-5. PMID: 9014149
- Rat and human hippocampal alpha5 subunit-containing gamma-aminobutyric AcidA receptors have alpha5 beta3 gamma2 pharmacological characteristics. | Sur, C., et al. 1998. Mol Pharmacol. 54: 928-33. PMID: 9804628
Inhibitor of:
ADAMTS-10.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
L-655,708, 5 mg | sc-204040 | 5 mg | $122.00 | |||
L-655,708, 25 mg | sc-204040A | 25 mg | $532.00 |
