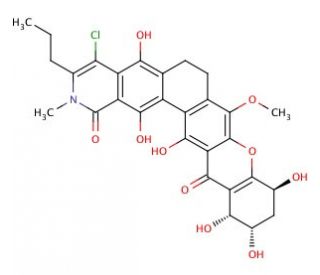
Kibdelone C (CAS 934464-79-6)
QUICK LINKS
Kibdelone C is a polyketide natural product that is studied for its interesting biological activities. In research, it is primarily investigated for its potential as a lead compound in the synthesis of new molecules with biologically relevant properties. Kibdelone C′s complex molecular architecture, characterized by multiple rings and stereocenters, provides a challenging and informative template for synthetic organic chemists. Its role in the study of structure-activity relationships is also significant, as modifications to its structure can lead to variations in biological activity, which are valuable for understanding molecular interactions at a fundamental level. Additionally, Kibdelone C is used in chemical biology studies aimed at elucidating the mode of action of similar polyketide compounds, contributing to the broader comprehension of how these molecules exert their effects at the cellular level.
Kibdelone C (CAS 934464-79-6) References
- Synthesis and biological evaluation of ABCD ring fragments of the kibdelones. | Sloman, DL., et al. 2011. Angew Chem Int Ed Engl. 50: 2511-5. PMID: 21370327
- Total synthesis and absolute stereochemical assignment of kibdelone C. | Sloman, DL., et al. 2011. J Am Chem Soc. 133: 9952-5. PMID: 21648477
- Enantioselective total synthesis of (-)-kibdelone C. | Butler, JR., et al. 2011. J Am Chem Soc. 133: 9956-9. PMID: 21648478
- Toluene dioxygenase mediated oxidation of halogen-substituted benzoate esters. | Semak, V., et al. 2012. Org Biomol Chem. 10: 4407-16. PMID: 22562668
- A radical access to highly functionalized tetrahydroxanthones. | Meister, AC., et al. 2013. Chemistry. 19: 10836-9. PMID: 23828509
- Dehydrogenative coupling to enable the enantioselective total synthesis of (-)-simaomicin α. | Wang, Y., et al. 2013. Angew Chem Int Ed Engl. 52: 10796-9. PMID: 24038677
- A photo-induced C-O bond formation methodology to construct tetrahydroxanthones. | Xiao, Z., et al. 2014. Chem Commun (Camb). 50: 5254-7. PMID: 24301299
- DMAP promoted tandem addition reactions forming substituted tetrahydroxanthones. | Castillo-Contreras, EB. and Dake, GR. 2014. Org Lett. 16: 1642-5. PMID: 24621113
- Synthesis and Biological Evaluation of Kibdelone C and Its Simplified Derivatives. | Rujirawanich, J., et al. 2016. J Am Chem Soc. 138: 10561-70. PMID: 27459345
- Enantioselective Halolactonization Reactions using BINOL-Derived Bifunctional Catalysts: Methodology, Diversification, and Applications. | Klosowski, DW., et al. 2018. J Org Chem. 83: 5954-5968. PMID: 29717607
- Convergent Synthesis of Kibdelone C. | Dai, Y., et al. 2018. Org Lett. 20: 2872-2875. PMID: 29738256
- Synthesis of N-alkylated 2-pyridones through Pummerer type reactions of activated sulfoxides and 2-fluoropyridine derivatives. | Hu, G., et al. 2018. Org Biomol Chem. 16: 4151-4158. PMID: 29785444
- Asymmetric Total Synthesis of the Complex Polycyclic Xanthone FD-594. | Xie, T., et al. 2020. Angew Chem Int Ed Engl. 59: 4360-4364. PMID: 31943607
- The Critical Role of 12-Methyl Group of Anthracycline Dutomycin to Its Antiproliferative Activity. | Xu, R., et al. 2022. Molecules. 27: PMID: 35630823
- Chemistry and biosynthesis of bacterial polycyclic xanthone natural products. | Kong, L., et al. 2022. Nat Prod Rep. 39: 2057-2095. PMID: 36083257
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Kibdelone C, 1 mg | sc-362759 | 1 mg | $280.00 |
