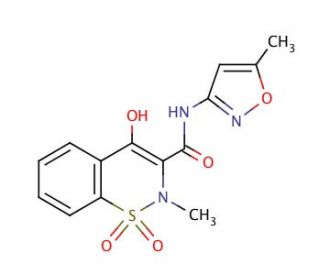
Isoxicam (CAS 34552-84-6)
QUICK LINKS
Isoxicam is a compound that is of particular interest in the field of biochemistry and enzyme inhibition studies. Its structure, which is characteristic of the oxicam class of compounds, makes it a focal point for research into the inhibition of certain enzymes. In particular, Isoxicam has been studied for its interaction with cyclooxygenase (COX) enzymes, which are involved in the biosynthesis of prostanoids, including prostaglandins, thromboxanes, and prostacyclins. Research involving Isoxicam may explore its potential to modulate the COX pathway, providing insights into the regulation of these physiologically active compounds. Investigations might also examine Isoxicam′s effects on inflammatory pathways at a molecular level. Furthermore, Isoxicam can serve as a lead compound in the design of new molecules with similar or improved properties, aiding in the study of molecular recognition and enzyme-substrate interactions.
Isoxicam (CAS 34552-84-6) References
- The anti=inflammatory properties of isoxicam (4-hydroxy-2methyl-N-(5-methyl-3isoxolyl-2H-1,2-benzothiazine-3-carboxamide 1,1-dioxide). | DiPasquale, G., et al. 1975. Agents Actions. 5: 256-63. PMID: 1236641
- Solid-phase molecularly imprinted pre-concentration and spectrophotometric determination of isoxicam in pharmaceuticals and human serum. | Rezaei, B., et al. 2009. Talanta. 78: 418-23. PMID: 19203603
- In vivo metabolism of isoxicam in rats, dogs, and monkeys. | Woolf, TF., et al. 1989. Drug Metab Dispos. 17: 662-8. PMID: 2575504
- In vitro metabolism of isoxicam by horseradish peroxidase. | Woolf, TF., et al. 1989. Xenobiotica. 19: 1369-77. PMID: 2618088
- Spectroscopic studies on the interaction of DNA with the copper complexes of NSAIDs lornoxicam and isoxicam. | Goswami, S., et al. 2016. Int J Biol Macromol. 93: 47-56. PMID: 27554932
- Painkiller Isoxicam and Its Copper Complex Can Form Inclusion Complexes with Different Cyclodextrins: A Fluorescence, Fourier Transform Infrared Spectroscopy, and Nuclear Magnetic Resonance Study. | Goswami, S., et al. 2017. J Phys Chem B. 121: 8454-8466. PMID: 28806512
- Experience with isoxicam and catabolin. | Sheppeard, H. and Couchman, KG. 1986. Br J Clin Pharmacol. 22 Suppl 2: 121S-124S. PMID: 3497659
- Lowering of plasma isoxicam concentrations with acetylsalicylic acid. | Grace, EM., et al. 1986. J Rheumatol. 13: 1119-21. PMID: 3560102
- The effect of isoxicam-aspirin combinations on the polyurethane sponge implantation model in the rat. | Garrett, R., et al. 1986. Br J Clin Pharmacol. 22 Suppl 2: 125S-128S. PMID: 3620271
- Fecal blood loss during isoxicam and piroxicam administration for 28 days. | Hooper, JW., et al. 1985. Clin Pharmacol Ther. 38: 533-7. PMID: 3876907
- Evaluation of the safety of isoxicam. | Burch, FX. 1985. Am J Med. 79: 28-32. PMID: 3904436
- The effect of administration of propranolol on the pharmacokinetics of isoxicam. | Caillé, G., et al. 1986. Biopharm Drug Dispos. 7: 63-9. PMID: 3955200
- Metabolic disposition of isoxicam in man, monkey, dog, and rat. | Borondy, PE. and Michniewicz, BM. 1984. Drug Metab Dispos. 12: 444-51. PMID: 6148211
- Isoxicam. | Downie, WW., et al. 1984. Clin Rheum Dis. 10: 385-99. PMID: 6391795
- Interaction of isoxicam with acetylsalicylic acid. | Esquivel, M., et al. 1984. Br J Clin Pharmacol. 18: 567-71. PMID: 6487496
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Isoxicam, 1 g | sc-235433 | 1 g | $99.00 |
