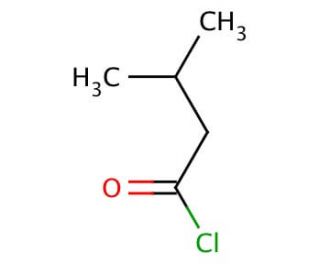
Molecular structure of Isovaleryl chloride, CAS Number: 108-12-3
Isovaleryl chloride (CAS 108-12-3)
Alternate Names:
3-Methylbutyryl chloride
Application:
Isovaleryl chloride is an acid halide and amino acid analog and potentially useful synthetic reagent
CAS Number:
108-12-3
Molecular Weight:
120.58
Molecular Formula:
C5H9ClO
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Isovaleryl chloride is a chlorinated derivative of isovaleric acid, utilized in various applications such as organic synthesis and laboratory experiments. Its applications encompass the study of free radicals′ effects on biological systems and the exploration of proteins′ structure and function. Moreover, Isovaleryl chloride serves as a potent inhibitor of the enzyme acetyl-CoA carboxylase, which is pivotal in fatty acid synthesis, resulting in reduced fatty acid production. Additionally, this compound has demonstrated inhibition of the enzyme dihydroxyacetone phosphate acyltransferase, a key player in glycerolipid synthesis.
Isovaleryl chloride (CAS 108-12-3) References
- A surprising mechanistic 'switch' in Lewis acid activation: a bifunctional, asymmetric approach to alpha-hydroxy acid derivatives. | Abraham, CJ., et al. 2008. J Am Chem Soc. 130: 17085-94. PMID: 19053448
- A catalytic asymmetric route to carbapenems. | Bodner, MJ., et al. 2009. Org Lett. 11: 3606-9. PMID: 19610642
- Novel and facile synthesis of furanodictines A and B based on transformation of 2-acetamido-2-deoxy-D-glucose into 3,6-anhydro hexofuranoses. | Ogata, M., et al. 2010. Carbohydr Res. 345: 230-4. PMID: 19962133
- Designing inhibitors against fructose 1,6-bisphosphatase: exploring natural products for novel inhibitor scaffolds. | Heng, S., et al. 2010. Eur J Med Chem. 45: 1478-84. PMID: 20116906
- Solid-phase synthesis and screening of N-acylated polyamine (NAPA) combinatorial libraries for protein binding. | Iera, JA., et al. 2010. Bioorg Med Chem Lett. 20: 6500-3. PMID: 20932761
- Synthesis, Compound, Emulsification, and Antibacterial Activity of Modified 1,2,4-Trizaole Derivatives. | Guo, H., et al. 2019. ACS Omega. 4: 9680-9685. PMID: 31460058
- Synthesis of legonmycins A and B, C(7a)-hydroxylated bacterial pyrrolizidines. | Lewis, WJM., et al. 2021. Beilstein J Org Chem. 17: 334-342. PMID: 33828615
- Inhibition of A549 Lung Cancer Cell Migration and Invasion by Ent-Caprolactin C via the Suppression of Transforming Growth Factor-β-Induced Epithelial-Mesenchymal Transition. | Kim, SY., et al. 2021. Mar Drugs. 19: PMID: 34436304
- Comprehensive exploration of chemical space using trisubstituted carboranes. | Asawa, Y., et al. 2021. Sci Rep. 11: 24101. PMID: 34916538
- Aminoacyl chain translocation catalysed by a type II thioesterase domain in an unusual non-ribosomal peptide synthetase. | Wang, S., et al. 2022. Nat Commun. 13: 62. PMID: 35013184
- Analysis of hyperforin (St. John's wort) action at TRPC6 channel leads to the development of a new class of antidepressant drugs. | El Hamdaoui, Y., et al. 2022. Mol Psychiatry. 27: 5070-5085. PMID: 36224261
- Genetic encoding of isobutyryl-, isovaleryl-, and β-hydroxybutryl-lysine in E. coli. | Christopher, JA., et al. 2022. RSC Adv. 12: 34142-34144. PMID: 36545614
- A push-pull strategy to control the western flower thrips, Frankliniella occidentalis, using alarm and aggregation pheromones. | Kim, CY., et al. 2023. PLoS One. 18: e0279646. PMID: 36827422
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Isovaleryl chloride, 25 g | sc-224028 | 25 g | $36.00 | |||
Isovaleryl chloride, 100 g | sc-224028A | 100 g | $61.00 |
