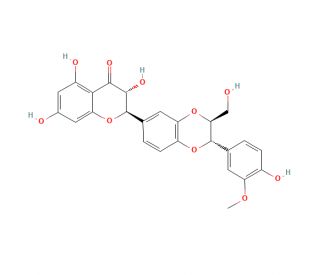
Isosilybin B (CAS 142796-22-3)
QUICK LINKS
Isosilybin B is a compound that functions as an antioxidant in experimental applications. It exerts its mechanism of action by scavenging free radicals and reducing oxidative stress at the cellular level. Isosilybin B may inhibit certain enzymes involved in the production of reactive oxygen species, thereby contributing to its antioxidant properties. It used to modulate various signaling pathways within cells, leading to the downregulation of inflammatory mediators and the upregulation of antioxidant enzymes. Isosilybin B also demonstrates the ability to chelate metal ions, further contributing to its antioxidant function. Isosilybin B plays a role in maintaining cellular homeostasis by counteracting oxidative damage and promoting a balanced redox state within cells.
Isosilybin B (CAS 142796-22-3) References
- Molecular structure and stereochemistry of silybin A, silybin B, isosilybin A, and isosilybin B, Isolated from Silybum marianum (milk thistle). | Lee, DY. and Liu, Y. 2003. J Nat Prod. 66: 1171-4. PMID: 14510591
- Milk thistle and prostate cancer: differential effects of pure flavonolignans from Silybum marianum on antiproliferative end points in human prostate carcinoma cells. | Davis-Searles, PR., et al. 2005. Cancer Res. 65: 4448-57. PMID: 15899838
- Isosilybin B and isosilybin A inhibit growth, induce G1 arrest and cause apoptosis in human prostate cancer LNCaP and 22Rv1 cells. | Deep, G., et al. 2007. Carcinogenesis. 28: 1533-42. PMID: 17389612
- Isosilybin B causes androgen receptor degradation in human prostate carcinoma cells via PI3K-Akt-Mdm2-mediated pathway. | Deep, G., et al. 2008. Oncogene. 27: 3986-98. PMID: 18332867
- Identifying the differential effects of silymarin constituents on cell growth and cell cycle regulatory molecules in human prostate cancer cells. | Deep, G., et al. 2008. Int J Cancer. 123: 41-50. PMID: 18435416
- HiFSA fingerprinting applied to isomers with near-identical NMR spectra: the silybin/isosilybin case. | Napolitano, JG., et al. 2013. J Org Chem. 78: 2827-39. PMID: 23461697
- An assessment of pharmacokinetics and antioxidant activity of free silymarin flavonolignans in healthy volunteers: a dose escalation study. | Zhu, HJ., et al. 2013. Drug Metab Dispos. 41: 1679-85. PMID: 23835761
- Silymarin Constituents Enhance ABCA1 Expression in THP-1 Macrophages. | Wang, L., et al. 2015. Molecules. 21: E55. PMID: 26729088
- The effect of milk thistle (Silybum marianum) and its main flavonolignans on CYP2C8 enzyme activity in human liver microsomes. | Albassam, AA., et al. 2017. Chem Biol Interact. 271: 24-29. PMID: 28457856
- Metabolism of flavonolignans in human hepatocytes. | Vrba, J., et al. 2018. J Pharm Biomed Anal. 152: 94-101. PMID: 29414024
- Identification of Human Sulfotransferases Active towards Silymarin Flavonolignans and Taxifolin. | Vrba, J., et al. 2020. Metabolites. 10: PMID: 32806559
- Recapitulation of Evidence of Phytochemical, Pharmacokinetic and Biomedical Application of Silybin. | Islam, A., et al. 2021. Drug Res (Stuttg). 71: 489-503. PMID: 34318464
- Development of a capillary electrophoresis method for the separation of flavonolignans in silymarin complex. | Riasová, P., et al. 2022. Electrophoresis. 43: 930-938. PMID: 34751959
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Isosilybin B, 5 mg | sc-506053 | 5 mg | $750.00 |
