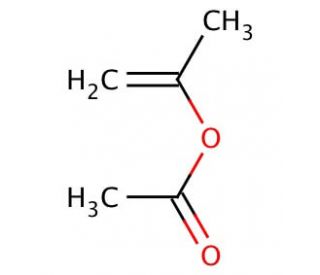
Molecular structure of Isopropenyl acetate
Isopropenyl acetate (CAS 108-22-5)
Alternate Names:
1-Methylvinyl acetate
Application:
Isopropenyl acetate is a biochemical used for proteomics research
CAS Number:
108-22-5
Molecular Weight:
100.12
Molecular Formula:
C5H8O2
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Isopropenyl acetate is a biochemical used for proteomics research. Isopropenyl acetate forms copolyperoxides of different compositions with indene by the free radical-initiated oxidative copolymerization. In industrial applications, isopropenyl acetate is often used as a solvent and intermediate in the synthesis of other chemical compounds. Its effectiveness as a solvent stems from its ability to dissolve a wide range of organic materials. This property is particularly useful in formulations where a fast-evaporating solvent is required.
Isopropenyl acetate (CAS 108-22-5) References
- Metal-catalyzed acyl transfer reactions of enol esters: role of Y5(OiPr)13O and (thd)2Y(OiPr) as transesterification catalysts. | Lin, MH. and RajanBabu, TV. 2000. Org Lett. 2: 997-1000. PMID: 10768206
- Lipase-catalyzed irreversible transesterification of 1-(2-furyl)ethanol using isopropenyl acetate. | Ghanem, A. and Schurig, V. 2001. Chirality. 13: 118-23. PMID: 11170255
- An Efficient Acylation of Tertiary Alcohols with Isopropenyl Acetate Mediated by an Oxime Ester and Cp(2)Sm(thf)(2). | Tashiro, D., et al. 1997. J Org Chem. 62: 8141-8144. PMID: 11671923
- Efficient lipase-catalyzed enantioselective desymmetrization of prochiral 2,2-disubstituted 1,3-propanediols and meso 1,2-diols using 1-ethoxyvinyl 2-furoate. | Akai, S., et al. 2002. J Org Chem. 67: 411-9. PMID: 11798311
- Enol esters: versatile substrates for Mannich-type multicomponent reactions. | Isambert, N., et al. 2007. Org Lett. 9: 4199-202. PMID: 17867693
- Direct synthesis of anti-1,3-diols through nonclassical reaction of aryl Grignard reagents with isopropenyl acetate. | Jiao, Y., et al. 2011. Org Lett. 13: 180-3. PMID: 21142039
- Per-O-acetylation of cellulose in dimethyl sulfoxide with catalyzed transesterification. | Chen, CY., et al. 2014. J Agric Food Chem. 62: 3446-52. PMID: 24678805
- UV resistance and dimensional stability of wood modified with isopropenyl acetate. | Nagarajappa, GB. and Pandey, KK. 2016. J Photochem Photobiol B. 155: 20-7. PMID: 26722999
- Lipase-catalyzed kinetic resolution as key step in the synthesis of enantiomerically pure σ ligands with 2-benzopyran structure. | Knappmann, I., et al. 2017. Bioorg Med Chem. 25: 3384-3395. PMID: 28501431
- Biocatalytic Friedel-Crafts Acylation and Fries Reaction. | Schmidt, NG., et al. 2017. Angew Chem Int Ed Engl. 56: 7615-7619. PMID: 28544673
- A New Family of Renewable Thermosets: Kraft Lignin Poly-adipates. | Di Francesco, D., et al. 2022. ChemSusChem. 15: e202200326. PMID: 35312238
- A Stoichiometric Solvent-Free Protocol for Acetylation Reactions. | Valentini, F., et al. 2022. Front Chem. 10: 842190. PMID: 35355791
- Cellulose triacetate synthesis via one-pot organocatalytic transesterification and delignification of pretreated bagasse. | Suzuki, S., et al. 2018. RSC Adv. 8: 21768-21776. PMID: 35541740
- Dimethyl Sulfoxide-Assisted, Iodine- and Ascorbic Acid-Catalyzed One-Pot Synthetic Approach for Constructing Highly Substituted Pyrazolo[1,5-a]quinoline Thioether Derivatives. | Saritha, R., et al. 2022. J Org Chem. 87: 13856-13872. PMID: 36215433
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Isopropenyl acetate, 250 ml | sc-396799 | 250 ml | $41.00 | |||
Isopropenyl acetate, 1 L | sc-396799A | 1 L | $108.00 |
