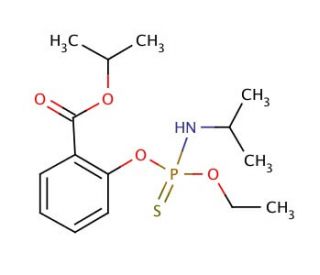
Isofenphos (CAS 25311-71-1)
QUICK LINKS
Isofenphos is an organophosphorus compound that has been used as a pesticide in agricultural practices. Isofenphos is known for its insecticidal properties, specifically targeting a wide range of pests, including insects, nematodes, and mites. This chemical acts by inhibiting the activity of the enzyme acetylcholinesterase, which is for the proper functioning of the nervous system in pests. By disrupting this enzyme, isofenphos interferes with the transmission of nerve impulses, leading to paralysis and ultimately the death of the target pests. Isofenphos has been employed in various agricultural settings to control pests in crops such as potatoes, soybeans, cotton, and fruits. Its effectiveness against a broad spectrum of pests has made it useful in pest management strategies.
Isofenphos (CAS 25311-71-1) References
- Blood cholinesterases as human biomarkers of organophosphorus pesticide exposure. | Nigg, HN. and Knaak, JB. 2000. Rev Environ Contam Toxicol. 163: 29-111. PMID: 10771584
- Isofenphos induced metabolic changes in K562 myeloid blast cells. | Boros, LG. and Williams, RD. 2001. Leuk Res. 25: 883-90. PMID: 11532522
- The relationship between isofenphos cholinergic toxicity and the development of polyneuropathy in hens and humans. | Moretto, A. and Lotti, M. 2002. Arch Toxicol. 76: 367-75. PMID: 12107655
- In vivo and in vitro percutaneous absorption and skin evaporation of isofenphos in man. | Wester, RC., et al. 1992. Fundam Appl Toxicol. 19: 521-6. PMID: 1426710
- Safety/risk assessment of pesticides: principles, procedures and examples. | Lu, FC. and Dourson, ML. 1992. Toxicol Lett. 64-65 Spec No: 783-7. PMID: 1471237
- Neurotoxicity of acute and repeated treatments of tabun, paraoxon, diisopropyl fluorophosphate and isofenphos to the hen. | Henderson, JD., et al. 1992. Toxicology. 72: 117-29. PMID: 1566275
- Organophosphate-induced delayed polyneuropathy. | Lotti, M. and Moretto, A. 2005. Toxicol Rev. 24: 37-49. PMID: 16042503
- Separation of organophosphorus pesticides by using nano-liquid chromatography. | Buonasera, K., et al. 2009. J Chromatogr A. 1216: 3970-6. PMID: 19321170
- Fenamiphos and related organophosphorus pesticides: environmental fate and toxicology. | Cáceres, T., et al. 2010. Rev Environ Contam Toxicol. 205: 117-62. PMID: 20044796
- A comparison of the persistence in a clay loam of single and repeated annual applications of seven granular insecticides used for corn rootworm control. | Harris, CR., et al. 1988. J Environ Sci Health B. 23: 1-32. PMID: 3372939
- Isofenphos and an in vitro activation assay for delayed neuropathic potential. | Chow, E., et al. 1986. Toxicol Appl Pharmacol. 83: 178-83. PMID: 3952746
- Toxicity of organophosphorus esters to laying hens after oral and dermal administration. | Francis, BM., et al. 1985. J Environ Sci Health B. 20: 73-95. PMID: 3989223
- Protective effect of pralidoxime on muscle fiber necrosis induced by organophosphate compounds. | Cavaliere, MJ., et al. 1998. J Toxicol Clin Toxicol. 36: 295-300. PMID: 9711194
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Isofenphos, 100 mg | sc-257626 | 100 mg | $250.00 |
