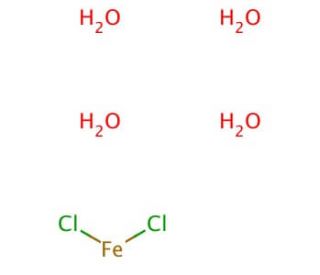
Molecular structure of Iron(II) chloride tetrahydrate, CAS Number: 13478-10-9
Iron(II) chloride tetrahydrate (CAS 13478-10-9)
CAS Number:
13478-10-9
Purity:
98%
Molecular Weight:
198.81
Molecular Formula:
FeCl2•4H2O
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Additionally, it contributes to the creation of metal complexes, like iron-chloride complexes. It also aids in the synthesis of catalysts and serves as a reagent in biochemistry, physiology, and biotechnology. This compound is characterized by its greenish crystalline appearance and is highly soluble in water, facilitating a variety of aqueous reactions. The presence of iron(II) or ferrous ion in this compound plays a role in redox reactions, serving as both an oxidizing and reducing agent depending on the environmental conditions. The tetrahydrate form stabilizes the iron(II) ions, preventing their rapid oxidation in air, which is a common challenge with ferrous compounds.
Iron(II) chloride tetrahydrate (CAS 13478-10-9) References
- Understanding the role of iron chlorides in the de novo synthesis of polychlorinated dibenzo-p-dioxins/dibenzofurans. | Ryan, SP. and Altwicker, ER. 2004. Environ Sci Technol. 38: 1708-17. PMID: 15074679
- Bis(2-pyridylimino)isoindolato iron(II) and cobalt(II) complexes: structural chemistry and paramagnetic NMR spectroscopy. | Kruck, M., et al. 2011. Dalton Trans. 40: 10406-15. PMID: 21687900
- In Situ Synthesis of Fe3O4 Nanoparticles and Wood Composite Properties of Three Tropical Species. | Moya, R., et al. 2022. Materials (Basel). 15: PMID: 35591726
- Nanocomposite Based on HA/PVTMS/Cl2FeH8O4 as a Gas and Temperature Sensor. | Nasiri, S., et al. 2022. Sensors (Basel). 22: PMID: 36560381
- Role of molecular strain on the solid-state synthesis of coordination compounds from iron(II) chloride tetrahydrate and 1,10-phenanthroline under mechanical stressRôle de la déformation moléculaire dans la synthèse de composés de coordination, à partir de poudres cristallines de dichlorure de fer tétrahydraté et de 1,10-phénanthroline soumis à une contrainte mécanique | Tadashi Ohshita, Daisuke Nakajima, Atsushi Tsukamoto, Naoko Tsuchiya, Tetsuhiko Isobe, Mamoru Senna, Naoki Yoshioka, Hidenari Inoue. 2002. Annales de Chimie Science des Matériaux. 27: 91-101.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Iron(II) chloride tetrahydrate, 50 g | sc-250168 | 50 g | $72.00 | |||
Iron(II) chloride tetrahydrate, 250 g | sc-250168A | 250 g | $109.00 |
