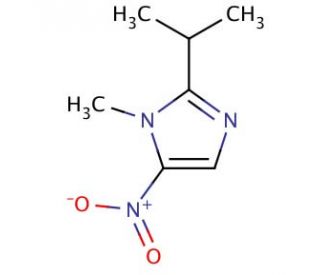
Ipropran (CAS 14885-29-1)
QUICK LINKS
Ipropran, a chemical primarily utilized in the study of cholinergic neurotransmission, acts as a cholinesterase inhibitor. This action is pivotal in research focused on understanding the dynamics of acetylcholine in the synaptic cleft, where ipropran inhibits the breakdown of acetylcholine, thereby prolonging its action at cholinergic synapses. This property makes it an essential tool for studying neuronal communication and the regulation of neurotransmitter levels. Research involving ipropran helps explain the mechanisms underlying cholinergic signaling pathways and their impact on cognitive and neural function. Moreover, its role extends to investigating potential protective strategies against neurotoxic agents that target cholinergic systems, offering insights into the development of neuroprotective models. By enhancing our understanding of neurotransmitter regulation and synaptic efficiency, ipropran contributes significantly to the field of neurobiology and pharmacology, aiding in the broader comprehension of neuronal communication and its implications for various biological processes and behaviors. Through these applications, ipropran advances scientific knowledge in neural system function and the modulation of cholinergic activity.
Ipropran (CAS 14885-29-1) References
- Effects of metronidazole, ipronidazole, and dibromochloropropane on rabbit and human sperm motility and fertility. | Foote, RH. 2002. Reprod Toxicol. 16: 749-55. PMID: 12401502
- Transfer of nitroimidazoles from contaminated beeswax to honey. | Mitrowska, K. and Antczak, M. 2017. Food Addit Contam Part A Chem Anal Control Expo Risk Assess. 34: 573-581. PMID: 27838954
- Determination of dimetridazole and ipronidazole in feeds at cross-contamination levels. | Hughes, DD. 1988. J Assoc Off Anal Chem. 71: 474-7. PMID: 3391941
- Treatment of bovine trichomoniasis with ipronidazole. | Skirrow, SZ. and Bondurant, RH. 1988. Aust Vet J. 65: 156. PMID: 3401163
- Metabolomics Approach on Non-Targeted Screening of 50 PPCPs in Lettuce and Maize. | Xue, W., et al. 2022. Molecules. 27: PMID: 35897888
- Efficacy of ipronidazole against trichomoniasis in beef bulls. | Skirrow, S., et al. 1985. J Am Vet Med Assoc. 187: 405-7. PMID: 4030475
- Chemotherapeutic, nutritional and biochemical compatibility of rofenaid and ipropran in turkeys. | Mitrovic, M., et al. 1972. Poult Sci. 51: 764-70. PMID: 4674999
- Residue analysis of ipronidazole and its metabolite at the 2 ppb level in turkey tissue. | MacDonald, A., et al. 1971. J Agric Food Chem. 19: 1222-7. PMID: 5132653
- Ipronidazole, an antihistomonal agent, as a turkey growth promotant. | Marusich, WL., et al. 1970. Poult Sci. 49: 98-101. PMID: 5462255
- Ipronidazole in the drinking water for treatment and prevention of experimental swine dysentery. | Olson, LD. 1977. Am J Vet Res. 38: 1289-92. PMID: 921022
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ipropran, 10 mg | sc-396425 | 10 mg | $396.00 |
