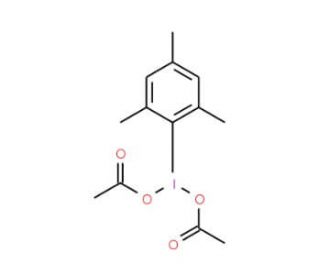
Iodomesitylene Diacetate (CAS 33035-41-5)
QUICK LINKS
Iodomesitylene Diacetate, referred to as TDI, holds significant prominence in scientific research applications. Iodomesitylene Diacetate serves as a intermediate in the synthesis of various compounds and has found extensive utilization as a reagent in the production of agrochemicals, and dyes. The applications of Iodomesitylene Diacetate in scientific research are diverse and encompass several domains. Furthermore, Iodomesitylene Diacetate has played a role in the synthesis of polymers, notably contributing to the formation of polyurethanes and polyesters. Additionally, its catalytic properties have been harnessed in the synthesis of heterocyclic compounds. Iodomesitylene Diacetate functions as an electrophilic reagent during the synthesis of various compounds. It readily engages with nucleophiles such as alcohols, amines, and carboxylic acids, initiating a reaction that culminates in the formation of a product. The process commences with the generation of a carbocation, which subsequently undergoes reaction with the nucleophile to yield the desired product.
Iodomesitylene Diacetate (CAS 33035-41-5) References
- Remarkably high reactivity of Pd(OAc)2/pyridine catalysts: nondirected C-H oxygenation of arenes. | Emmert, MH., et al. 2011. Angew Chem Int Ed Engl. 50: 9409-12. PMID: 21948437
- Base Mediated Synthesis of Alkyl-aryl Ethers from the Reaction of Aliphatic Alcohols and Unsymmetric Diaryliodonium Salts. | Sundalam, SK. and Stuart, DR. 2015. J Org Chem. 80: 6456-66. PMID: 26020831
- Arylation with Diaryliodonium Salts. | Olofsson, B. 2015. Top Curr Chem.. PMID: 26499897
- Unsymmetrical Aryl(2,4,6-trimethoxyphenyl)iodonium Salts: One-Pot Synthesis, Scope, Stability, and Synthetic Studies. | Seidl, TL., et al. 2016. J Org Chem. 81: 1998-2009. PMID: 26828570
- Decarboxylative sp3 C-N coupling via dual copper and photoredox catalysis. | Liang, Y., et al. 2018. Nature. 559: 83-88. PMID: 29925943
- Mechanistic Study on the Decarboxylative sp3 C-N Cross-Coupling between Alkyl Carboxylic Acids and Nitrogen Nucleophiles via Dual Copper and Photoredox Catalysis. | Zhao, X., et al. 2019. Inorg Chem. 58: 12669-12677. PMID: 31498616
- Effect of the Ligand Backbone on the Reactivity and Mechanistic Paradigm of Non-Heme Iron(IV)-Oxo during Olefin Epoxidation. | Biswas, JP., et al. 2021. Angew Chem Int Ed Engl. 60: 14030-14039. PMID: 33836110
- Metallaphotoredox: The Merger of Photoredox and Transition Metal Catalysis. | Chan, AY., et al. 2022. Chem Rev. 122: 1485-1542. PMID: 34793128
- Nontraditional Fragment Couplings of Alcohols and Carboxylic Acids: C(sp3)-C(sp3) Cross-Coupling via Radical Sorting. | Sakai, HA. and MacMillan, DWC. 2022. J Am Chem Soc. 144: 6185-6192. PMID: 35353531
- Recent developments in decarboxylative cross-coupling reactions between carboxylic acids and N-H compounds. | Arshadi, S., et al. 2019. RSC Adv. 9: 8964-8976. PMID: 35517670
- Nickel Catalysis via SH2 Homolytic Substitution: The Double Decarboxylative Cross-Coupling of Aliphatic Acids. | Tsymbal, AV., et al. 2022. J Am Chem Soc. 144: 21278-21286. PMID: 36375080
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Iodomesitylene Diacetate, 5 g | sc-295187 | 5 g | $213.00 | |||
Iodomesitylene Diacetate, 10 g | sc-295187A | 10 g | $379.00 |
