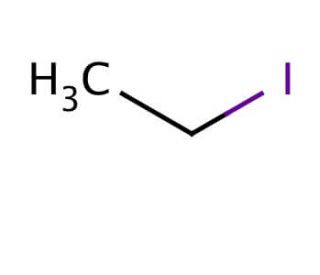
Iodoethane (CAS 75-03-6)
QUICK LINKS
Iodoethane, also known as ethyl iodide, is a versatile organic compound. This colorless and volatile liquid serves primarily as a precursor for synthesizing various organic compounds. It also finds utility as a reagent, solvent, and catalyst in laboratory experiments. However, it is important to note that iodoethane poses hazards due to its flammability and low boiling point, requiring careful handling. The mechanism of action of iodoethane relies on its capacity to engage in substitution reactions. As a nucleophile, it readily donates electrons and reacts with electrophiles, including alkyl halides, to generate novel compounds. This characteristic enables iodoethane to participate actively in chemical transformations.
Iodoethane (CAS 75-03-6) References
- Reductive dechlorination of tetrachloroethene to ethene by a two-component enzyme pathway. | Magnuson, JK., et al. 1998. Appl Environ Microbiol. 64: 1270-5. PMID: 10671186
- The quaternisation reaction of phosphines and amines in aliphatic alcohols. A similarity analysis using the isokinetic, isosolvent and isoselective relationships. | Pinheiro, LM., et al. 2004. Org Biomol Chem. 2: 1330-8. PMID: 15105923
- Rates and mechanisms for the reactions of chlorine atoms with iodoethane and 2-iodopropane. | Orlando, JJ., et al. 2005. J Phys Chem A. 109: 6659-75. PMID: 16834018
- Methimazole-based ionic liquids. | Siriwardana, AI., et al. 2008. J Org Chem. 73: 4676-9. PMID: 18402480
- Microwave synthesis of near infrared heptamethine cyanine dye. | Winstead, AJ., et al. 2008. J Microw Power Electromagn Energy. 42: 35-41. PMID: 19227081
- Oxidation of monohalogenated ethanes and n-chlorinated alkanes by whole cells of Nitrosomonas europaea. | Rasche, ME., et al. 1990. J Bacteriol. 172: 5368-73. PMID: 2394686
- An efficient buffer-mediated control between free radical substitution and proton-coupled electron transfer: dehalogenation of iodoethane by the α-hydroxyethyl radical in aqueous solution. | Ljubić, I., et al. 2013. Phys Chem Chem Phys. 15: 18001-11. PMID: 24061544
- Synthesis of amphiphilic 6-carboxypullulan ethers. | Pereira, JM., et al. 2014. Carbohydr Polym. 100: 65-73. PMID: 24188839
- Synthesis, Crystal Structure, and Biological Activity of Ethyl 4-Methyl-2,2-dioxo-1H-2λ⁶,1-benzothiazine-3-carboxylate Polymorphic Forms. | Ukrainets, IV., et al. 2018. Sci Pharm. 86: PMID: 29848976
- Subpicosecond HI elimination in the 266 nm photodissociation of branched iodoalkanes. | Todt, MA., et al. 2020. Phys Chem Chem Phys. 22: 27338-27347. PMID: 33231219
- Responsive Quaternized PDMAEMA Copolymers with Antimicrobial Action. | Manouras, T., et al. 2021. Polymers (Basel). 13: PMID: 34577950
- Synthesis, structure and reactivity of μ3-SnH capped trinuclear nickel cluster. | Torquato, NA., et al. 2022. Chem Sci. 13: 11382-11387. PMID: 36320577
- N-alkylation of 17-azasteroids. | Morzycki, JW. and Lotowski, Z. 1994. Steroids. 59: 30-3. PMID: 8140599
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Iodoethane, 5 g | sc-215184 | 5 g | $38.00 | |||
Iodoethane, 100 g | sc-215184A | 100 g | $59.00 |
