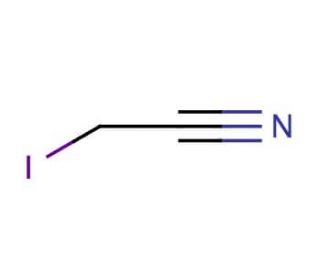
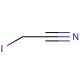
Iodoacetonitrile (CAS 624-75-9)
QUICK LINKS
Iodoacetonitrile is extensively researched within chemical synthesis, particularly for its role as an alkylating agent in the formation of carbon-nitrogen bonds. This compound is pivotal in studies involving nucleophilic substitution reactions where it acts as a versatile precursor for the synthesis of various organic compounds, including agrochemicals. The reactivity of Iodoacetonitrile under different conditions is a key area of investigation, as it influences the efficiency and selectivity of these reactions. Researchers also focus on the mechanisms by which Iodoacetonitrile interacts with other molecules, aiming to elucidate the pathways and improve reaction outcomes. Additionally, the stability and handling safety of Iodoacetonitrile are examined to optimize its use in laboratory and industrial settings.
Iodoacetonitrile (CAS 624-75-9) References
- Indium- or Zinc-Mediated One-Pot Synthesis of Homoallylamines, beta-Amino Esters, and beta-Amino Nitriles. | Choucair, B., et al. 2000. Org Lett. 2: 1851-1853. PMID: 10891174
- Haloacetonitriles vs. regulated haloacetic acids: are nitrogen-containing DBPs more toxic? | Muellner, MG., et al. 2007. Environ Sci Technol. 41: 645-51. PMID: 17310735
- Intermolecular nonreductive alkylation of enamides via radical-polar crossover. | Friestad, GK. and Wu, Y. 2009. Org Lett. 11: 819-22. PMID: 19199766
- Occupational erythema multiforme-like contact reaction to iodoacetonitrile in a chemistry student. | Foti, C., et al. 2011. Contact Dermatitis. 64: 180-1. PMID: 21272032
- Toxicity of drinking water disinfection byproducts: cell cycle alterations induced by the monohaloacetonitriles. | Komaki, Y., et al. 2014. Environ Sci Technol. 48: 11662-9. PMID: 25185076
- Cyanoacetic Acid as a Masked Electrophile: Transition-Metal-Free Cyanomethylation of Amines and Carboxylic Acids. | Wang, H., et al. 2015. Chemistry. 21: 18333-7. PMID: 26511114
- Visible-Light-Induced Regioselective Cyanomethylation of Imidazopyridines and Its Application in Drug Synthesis. | Chang, Q., et al. 2017. J Org Chem. 82: 5391-5397. PMID: 28470069
- Chlorination of Source Water Containing Iodinated X-ray Contrast Media: Mutagenicity and Identification of New Iodinated Disinfection Byproducts. | Postigo, C., et al. 2018. Environ Sci Technol. 52: 13047-13056. PMID: 30339747
- Comparative Quantitative Toxicology and QSAR Modeling of the Haloacetonitriles: Forcing Agents of Water Disinfection Byproduct Toxicity. | Wei, X., et al. 2020. Environ Sci Technol. 54: 8909-8918. PMID: 32551543
- Monohaloacetonitriles induce cytotoxicity and exhibit different mode of action in endocrine disruption. | Park, CG., et al. 2021. Sci Total Environ. 761: 143316. PMID: 33190885
- Formation and control of disinfection by-products from iodinated contrast media attenuation through sequential treatment processes of ozone-low pressure ultraviolet light followed by chlorination. | Lopez-Prieto, IJ., et al. 2021. Chemosphere. 278: 130394. PMID: 34126675
- Diastereoselective palladium-catalyzed C(sp3)-H cyanomethylation of amino acid and carboxylic acid derivatives. | Garai, S., et al. 2022. Chem Commun (Camb). 58: 7793-7796. PMID: 35735087
- NRF2-ARE signaling is responsive to haloacetonitrile-induced oxidative stress in human keratinocytes. | Xue, P., et al. 2022. Toxicol Appl Pharmacol. 450: 116163. PMID: 35842135
- Overlooked Iodo-Disinfection Byproduct Formation When Cooking Pasta with Iodized Table Salt. | Dong, H., et al. 2023. Environ Sci Technol. 57: 3538-3548. PMID: 36802504
- Synthesis and antiviral activity of 5-[(cyanomethylene)oxy]-2'-deoxyuridine. | Huang, GF., et al. 1981. J Med Chem. 24: 390-3. PMID: 6267280
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Iodoacetonitrile, 10 g | sc-250160 | 10 g | $92.00 |
