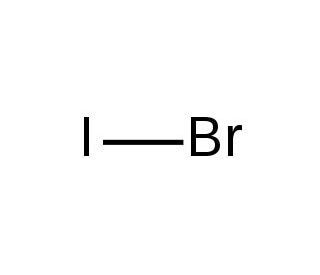
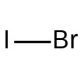
Iodine monobromide (CAS 7789-33-5)
QUICK LINKS
Iodine monobromide (IMB) is an inorganic compound comprising one iodine atom and one bromine atom. This colorless, volatile solid exhibits slight solubility in water. In the realm of scientific research, Iodine monobromide′s versatility shines through various applications. It has been widely employed as a reagent in synthesizing organic compounds, particularly bromoiodoacetals, which prove indispensable as intermediates in organic synthesis. Additionally, Iodine monobromide plays a role in the synthesis of bromoiodoacetates, known for their catalytic properties in organic reactions. The compound also serves as a catalyst in polymer synthesis, contributing to the development of novel materials. Notably, the preparation of bromoiodoacetamide, facilitated by Iodine monobromide, emerges as a valuable reagent in peptide synthesis. Iodine monobromide demonstrates intriguing properties, with the bromine atom acting as a Lewis acid, capable of accepting electrons from other molecules. This fascinating behavior enables Iodine monobromide to form complexes with various molecules, subsequently serving as catalysts in organic reactions. Such catalytic capabilities make Iodine monobromide a valuable asset in the realm of chemical transformations, offering opportunities for enhancing the efficiency and selectivity of organic processes.
Iodine monobromide (CAS 7789-33-5) References
- Products of the determination of the iodine value with iodine monobromide. | Imming, P. and Germershaus, O. 2002. Arch Pharm (Weinheim). 335: 449-51. PMID: 12447919
- Electrophile-induced ether transfer: a new approach to polyketide structural units. | Liu, K., et al. 2006. Org Lett. 8: 5393-5. PMID: 17078726
- Iodine monobromide catalysed regioselective synthesis of 3-arylquinolines from α-aminoacetophenones and trans-β-nitrostyrenes. | Gattu, R., et al. 2019. Org Biomol Chem. 17: 347-353. PMID: 30548050
- Direct mapping of curve-crossing dynamics in IBr by attosecond transient absorption spectroscopy. | Kobayashi, Y., et al. 2019. Science. 365: 79-83. PMID: 31273121
- Direct field evidence of autocatalytic iodine release from atmospheric aerosol. | Tham, YJ., et al. 2021. Proc Natl Acad Sci U S A. 118: PMID: 33479177
- Ultraviolet spectrophotometric determination of phenols in natural and waste waters with iodine monobromide. | Bosch, F., et al. 1987. Analyst. 112: 1335-7. PMID: 3425935
- Synthesis, Structure, and UV-Vis Characterization of Antimony(III) Phthalocyanine: [(SbPc)2(Sb2I8)(SbBr3)]2. | Kubiak, R. and Janczak, J. 2022. Molecules. 27: PMID: 35335201
- Total Synthesis of (-)-Bastimolide A: A Showcase for Type I Anion Relay Chemistry. | Cox, JB., et al. 2022. Angew Chem Int Ed Engl. 61: e202204884. PMID: 35608327
- Comprehensive Characterization of Oils and Fats of Six Species from the Colombian Amazon Region with Industrial Potential. | Lozano-Garzón, K., et al. 2023. Biomolecules. 13: PMID: 37371565
- Crystal structures and Hirshfeld surface analyses of N,N-di-methyl-acetamide-1-(dimethyl-λ4-aza-nyl-idene)ethan-1-ol tribromide (1/1), N,N-di-methyl-acetamide-1-(dimethyl-λ4-aza-nyl-idene)ethan-1-ol di-bromido-iodate (1/1) and N,N-di-methyl-acetamide-1-(dimethyl-λ4-aza-nyl-idene)ethan-1-ol di-chlorido-iodate (1/1). | Mammadova, GZ., et al. 2023. Acta Crystallogr E Crystallogr Commun. 79: 690-697. PMID: 37601404
- Exploring the potentials of Ziziphus mauritiana Lam. seed kernel oil as pharmaceutical oil base: Physicochemical characterization and ketoconazole soap formulation. | Pandey, J., et al. 2023. Heliyon. 9: e21034. PMID: 38106666
- Comparative evaluation of physicochemical and antimicrobial properties of rubber seed oil from different regions of Bangladesh. | Alam, MA., et al. 2024. Heliyon. 10: e25544. PMID: 38384561
- An Alkyne-Bridged Covalent Organic Framework Featuring Interactive Pockets for Bromine Capture. | De, A., et al. 2024. Angew Chem Int Ed Engl. e202403658. PMID: 38738600
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Iodine monobromide, 25 g | sc-252907 | 25 g | $114.00 | |||
Iodine monobromide, 50 g | sc-252907A | 50 g | $156.00 |
