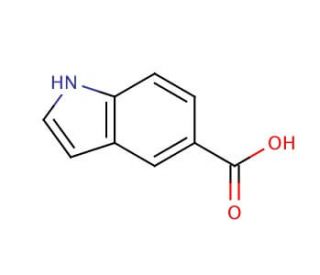
Indole-5-carboxylic acid (CAS 1670-81-1)
QUICK LINKS
Indole-5-carboxylic acid, or ICA, is a significant organic compound involved in numerous applications across scientific research and industrial sectors. This carboxylic acid, which originates from indole, is recognized for its varying physiological and biochemical impacts. Its use in scientific research is extensive, particularly in the synthesis of diverse compounds like indole-3-acetic acid and indole-3-butyric acid. Additionally, Indole-5-carboxylic acid plays a role in the synthesis of certain compounds like indomethacin and ibuprofen. It is also applied in the creation of various polymers, such as polyurethanes and polyesters. Although the exact mechanism of action of Indole-5-carboxylic acid remains unclear, it is known to interact with several receptors in the body, including the serotonin receptor and the gamma-aminobutyric acid (GABA) receptor. It is also reported to interact with various enzymes like cyclooxygenase-2 (COX-2) and 5-hydroxytryptamine (5-HT) receptors.
Indole-5-carboxylic acid (CAS 1670-81-1) References
- Electrochemical properties and Fourier transform-infrared spectroscopic investigations of the redox behaviour of poly(indole-5-carboxylic acid) in LiClO4-acetonitrile solutions. | Billaud, D., et al. 2003. Spectrochim Acta A Mol Biomol Spectrosc. 59: 163-8. PMID: 12509157
- Oligomerization of indole derivatives with incorporation of thiols. | Mutulis, F., et al. 2008. Molecules. 13: 1846-63. PMID: 18794789
- 1-(5-Carboxyindol-1-yl)propan-2-one inhibitors of human cytosolic phospholipase A2α: effect of substituents in position 3 of the indole scaffold on inhibitory potency, metabolic stability, solubility, and bioavailability. | Bovens, S., et al. 2010. J Med Chem. 53: 8298-308. PMID: 21067218
- Photophysics of indole-2-carboxylic acid (I2C) and indole-5-carboxylic acid (I5C): heavy atom effect. | Kowalska-Baron, A., et al. 2013. Spectrochim Acta A Mol Biomol Spectrosc. 116: 183-95. PMID: 23933843
- Convergent and enantioselective syntheses of cytosolic phospholipase A(2)α inhibiting N-(1-indazol-1-ylpropan-2-yl)carbamates. | Sundermann, T., et al. 2014. Org Biomol Chem. 12: 4021-30. PMID: 24817191
- A Label-Free Electrochemical Biosensor Based on a Reduced Graphene Oxide and Indole-5-Carboxylic Acid Nanocomposite for the Detection of Klebsiella pneumoniae. | Zhang, Z., et al. 2017. J AOAC Int. 100: 548-552. PMID: 28118564
- [Carboxyl-11 C]Labelling of Four High-Affinity cPLA2α Inhibitors and Their Evaluation as Radioligands in Mice by Positron Emission Tomography. | Fisher, MJ., et al. 2018. ChemMedChem. 13: 138-146. PMID: 29232493
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Indole-5-carboxylic acid, 1 g | sc-255216 | 1 g | $50.00 |
