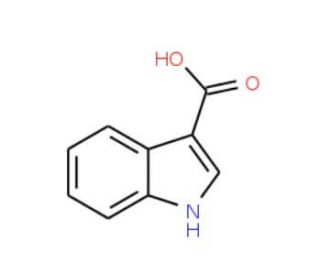
Molecular structure of Indole-3-carboxylic acid, CAS Number: 771-50-6
Indole-3-carboxylic acid (CAS 771-50-6)
See product citations (1)
Alternate Names:
β-Indolylcarboxylic acid
CAS Number:
771-50-6
Molecular Weight:
161.16
Molecular Formula:
C9H7NO2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Indole-3-carboxylic acid is a naturally occurring compound abundant in various plants and vegetables like broccoli, cabbage, and cauliflower. Scientific studies have examined its potential to influence enzyme and hormone activity, leading to possible anti-inflammatory effects. While the exact mechanism remains incompletely understood, it is thought to act by modulating enzymes and hormones, and even inducing apoptosis in specific cancer cell types.
Indole-3-carboxylic acid (CAS 771-50-6) References
- Formation of indole-3-carboxylic acid by Chromobacterium violaceum. | Davis, PJ., et al. 1976. J Bacteriol. 126: 544-6. PMID: 1262308
- The metabolism of indole-3-carboxylic acid by the rat. | ACHESON, RM. and KING, LJ. 1963. Biochim Biophys Acta. 71: 643-9. PMID: 14010848
- Metabolism of Tryptophan, Indole-3-acetic Acid, and Related Compounds in Parasitic Plants from the Genus Orobanche. | Magnus, V., et al. 1982. Plant Physiol. 69: 853-8. PMID: 16662308
- Production of indole-3-acetic acid and related indole derivatives from L-tryptophan by Rubrivivax benzoatilyticus JA2. | Mujahid, M., et al. 2011. Appl Microbiol Biotechnol. 89: 1001-8. PMID: 20972782
- Identification of indole-3-carboxylic acid as mediator of priming against Plectosphaerella cucumerina. | Gamir, J., et al. 2012. Plant Physiol Biochem. 61: 169-79. PMID: 23116603
- Identification of indole-3-carboxylic acids as non-ATP-competitive Polo-like kinase 1 (Plk1) inhibitors. | Liu, M., et al. 2015. Bioorg Med Chem Lett. 25: 431-4. PMID: 25556101
- Design, synthesis and preliminary biological evaluation of indole-3-carboxylic acid-based skeleton of Bcl-2/Mcl-1 dual inhibitors. | Liu, T., et al. 2017. Bioorg Med Chem. 25: 1939-1948. PMID: 28233676
- Energetic Effect of the Carboxylic Acid Functional Group in Indole Derivatives. | Carvalho, TMT., et al. 2017. J Phys Chem A. 121: 2980-2989. PMID: 28358511
- Starch degradation, abscisic acid and vesicular trafficking are important elements in callose priming by indole-3-carboxylic acid in response to Plectosphaerella cucumerina infection. | Gamir, J., et al. 2018. Plant J. 96: 518-531. PMID: 30051514
- Engineering of a chromogenic enzyme screening system based on an auxiliary indole-3-carboxylic acid monooxygenase. | Časaitė, V., et al. 2019. Microbiologyopen. 8: e00795. PMID: 30666828
- Accumulating evidences of callose priming by indole- 3- carboxylic acid in response to Plectospharella cucumerina. | Pastor-Fernández, J., et al. 2019. Plant Signal Behav. 14: 1608107. PMID: 31010375
- Ganoderma lucidum promotes sleep through a gut microbiota-dependent and serotonin-involved pathway in mice. | Yao, C., et al. 2021. Sci Rep. 11: 13660. PMID: 34211003
- Indole-3-Carboxylic Acid From the Endophytic Fungus Lasiodiplodia pseudotheobromae LPS-1 as a Synergist Enhancing the Antagonism of Jasmonic Acid Against Blumeria graminis on Wheat. | Que, Y., et al. 2022. Front Cell Infect Microbiol. 12: 898500. PMID: 35860382
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Indole-3-carboxylic acid, 5 g | sc-255214 | 5 g | $27.00 | |||
Indole-3-carboxylic acid, 25 g | sc-255214A | 25 g | $42.00 | |||
Indole-3-carboxylic acid, 100 g | sc-255214B | 100 g | $146.00 | |||
Indole-3-carboxylic acid, 250 g | sc-255214C | 250 g | $270.00 | |||
Indole-3-carboxylic acid, 1 kg | sc-255214D | 1 kg | $1022.00 |
