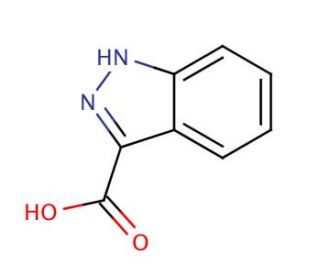
Molecular structure of Indazole-3-carboxylic Acid, CAS Number: 4498-67-3
Indazole-3-carboxylic Acid (CAS 4498-67-3)
See product citations (1)
Alternate Names:
1H-Indazole-3-carboxylic Acid; 3(1H)-Indazolecarboxylic Acid; 3-Carboxy-1H-indazole; 3-Carboxyindazole; NSC 520610
Application:
Indazole-3-carboxylic Acid is an antiinflammatory derivative of indole
CAS Number:
4498-67-3
Purity:
≥99%
Molecular Weight:
162.15
Molecular Formula:
C8H6N2O2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Derivative of indole that is useful in the treatment of pain and inflammation.
Indazole-3-carboxylic Acid (CAS 4498-67-3) References
- Characterization of the FAD-containing N-methyltryptophan oxidase from Escherichia coli. | Khanna, P. and Schuman Jorns, M. 2001. Biochemistry. 40: 1441-50. PMID: 11170472
- Structure-activity relationship study and discovery of indazole 3-carboxamides as calcium-release activated calcium channel blockers. | Bai, S., et al. 2017. Bioorg Med Chem Lett. 27: 393-397. PMID: 28057422
- Anti-cancer and anti-inflammatory activities of a new family of coordination compounds based on divalent transition metal ions and indazole-3-carboxylic acid. | García-Valdivia, AA., et al. 2021. J Inorg Biochem. 215: 111308. PMID: 33257004
- Structural and biochemical characterization of the prenylated flavin mononucleotide-dependent indole-3-carboxylic acid decarboxylase. | Gahloth, D., et al. 2022. J Biol Chem. 298: 101771. PMID: 35218772
- Indazoles as indole bioisosteres: synthesis and evaluation of the tropanyl ester and amide of indazole-3-carboxylate as antagonists at the serotonin 5HT3 receptor. | Fludzinski, P., et al. 1987. J Med Chem. 30: 1535-7. PMID: 3625701
- NIR and UV induced transformations of indazole-3-carboxylic acid isolated in low temperature matrices. | Pagacz-Kostrzewa, M., et al. 2023. Spectrochim Acta A Mol Biomol Spectrosc. 290: 122283. PMID: 36586171
- Effect of lonidamine derivatives on the inhibition of transformed cell area expansion. | Aoyama, M., et al. 2023. Biochem Biophys Rep. 34: 101480. PMID: 37180755
- Novel multi-target ligands of dopamine and serotonin receptors for the treatment of schizophrenia based on indazole and piperazine scaffolds-synthesis, biological activity, and structural evaluation. | Stępnicki, P., et al. 2023. J Enzyme Inhib Med Chem. 38: 2209828. PMID: 37184096
- Novel rhenium oxocomplexes of indazole-3-carboxylic acid – Synthesis, X-ray studies, spectroscopic characterization and DFT calculations | Machura, B., Świetlicka, A., Wolff, M., & Kruszynski, R. 2010. Polyhedron. 29(9): 2061-2069.
- Molecular modeling of indazole-3-carboxylic acid and its metal complexes (Zn, Ni, Co, Fe and Mn) as NO synthase inhibitors: DFT calculations, docking studies and molecular dynamics simulations | da Silva, T. U., da Silva, E. T., de Carvalho Pougy, K., da Silva Lima, C. H., & de Paula Machado, S. 2022. Inorganic Chemistry Communications. 135: 109120.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Indazole-3-carboxylic Acid, 2.5 g | sc-218595 | 2.5 g | $105.00 |
