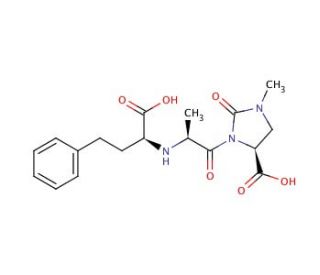
Molecular structure of Imidaprilat, CAS Number: 89371-44-8
Imidaprilat (CAS 89371-44-8)
Alternate Names:
(4S)-3-{(2S)-2-[N-((1S)-1-Carboxy-3-phenylpropyl)amino]propionyl}-1-methyl-2-oxoimidazolidine-4-carboxylic Acid; Imidapril Diacid
Application:
Imidaprilat is an ACE ihibitor
CAS Number:
89371-44-8
Molecular Weight:
377.39
Molecular Formula:
C18H23N3O6
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Imidaprilat facilitates the investigation of ion metabolism and transport, and it aids in analyzing how certain substances might interact with the immune system. It has a high affinity for and selectively inhibits certain enzymes, which is relevant in the exploration of enzyme behavior and kinetics.Functioning as a guanylate cyclase inhibitor, imidaprilat interacts with this enzyme, for blood pressure regulation, by hindering its ability to convert guanosine triphosphate (GTP) into cyclic guanosine monophosphate (cGMP).
Imidaprilat (CAS 89371-44-8) References
- Protective effect of imidaprilat, a new angiotensin-converting enzyme inhibitor against 1-methyl-4-phenylpyridinium ion-induced *OH generation in rat striatum. | Obata, T. 1999. Eur J Pharmacol. 378: 39-45. PMID: 10478563
- Protective effect of imidaprilat, an angiotensin-converting enzyme inhibitor on *OH generation in rat myocardium. | Obata, T. and Yamanaka, Y. 1999. Biochim Biophys Acta. 1472: 62-70. PMID: 10572926
- Determination of imidapril and imidaprilat in human plasma by high-performance liquid chromatography-electrospray ionization tandem mass spectrometry. | Mabuchi, M., et al. 1999. J Chromatogr B Biomed Sci Appl. 734: 145-53. PMID: 10574200
- Different inhibition of enalaprilat and imidaprilat on bradykinin metabolizing enzymes. | Sakamoto, K., et al. 2000. Life Sci. 67: 2159-65. PMID: 11045597
- Comparison of the effect of imidaprilat and ramiprilat on broncho-constriction and hypotension induced by bradykinin in guinea pigs. | Doi, H. and Narita, H. 2000. Inflamm Res. 49: 506-12. PMID: 11089901
- Protection of the cardiovascular system by imidapril, a versatile angiotensin-converting enzyme inhibitor. | Hosoya, K. and Ishimitsu, T. 2002. Cardiovasc Drug Rev. 20: 93-110. PMID: 12177688
- Imidaprilat suppresses nonylphenol and 1-methyl-4-phenylpyridinium ion (MPP+)-induced hydroxyl radical generation in rat striatum. | Obata, T. 2006. Neurosci Res. 54: 192-6. PMID: 16386813
- Imidaprilat, an angiotensin-converting enzyme inhibitor exerts neuroprotective effect via decreasing dopamine efflux and hydroxyl radical generation induced by bisphenol A and MPP+ in rat striatum. | Obata, T. 2006. Brain Res. 1071: 250-3. PMID: 16406270
- Imidaprilat inhibits matrix metalloproteinase-2 activity in human cardiac fibroblasts induced by interleukin-1beta via NO-dependent pathway. | Guo, XG., et al. 2008. Int J Cardiol. 126: 414-20. PMID: 18192036
- MMP-9 inhibition by ACE inhibitor reduces oxidized LDL-mediated foam-cell formation. | Kojima, C., et al. 2010. J Atheroscler Thromb. 17: 97-105. PMID: 20093780
- Steady-state pharmacokinetics and pharmacodynamics of imidaprilat, an active metabolite of imidapril, a new angiotensin-converting enzyme inhibitor in spontaneously hypertensive rats. | Yamanaka, K., et al. 1996. J Pharm Sci. 85: 1234-7. PMID: 8923331
- Pharmacokinetic and pharmacodynamic study of imidaprilat, an active metabolite of imidapril, a new angiotensin-converting enzyme inhibitor, in spontaneously hypertensive rats. | Yamanaka, K., et al. 1997. J Pharm Biomed Anal. 15: 1851-9. PMID: 9278890
- Effect of .OH scavenging action by non-SH-containing angiotensin converting enzyme inhibitor imidaprilat using microdialysis. | Obata, T. and Yamanaka, Y. 1998. J Physiol Paris. 92: 1-4. PMID: 9638590
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Imidaprilat, 1 mg | sc-218591 | 1 mg | $500.00 |
