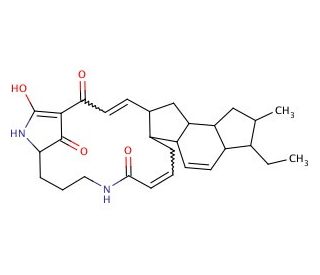
Molecular structure of Ikarugamycin, CAS Number: 36531-78-9
Ikarugamycin (CAS 36531-78-9)
See product citations (4)
Application:
Ikarugamycin is a macrolactam antibiotic effective gainst gram-positive bacteria
CAS Number:
36531-78-9
Purity:
≥95%
Molecular Weight:
478.6
Molecular Formula:
C29H38N2O4
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Ikarugamycin is an antiprotozoal macrolactam antibiotic demonstrating cytostatic effects and activity against gram-positive bacteria. It is described to inhibit clathrin-dependent endocytosis, as well as oxidized LDL-induced accumulation of cholesteryl ester in macrophage J774. Ikarugamycin exhibits cytotoxic effects in cancer cell lines, inhibiting cell proliferation (IC50 = 221.3 nM in HL-60 cells) through genotoxicity and by inducing apoptosis and activation of caspases. It also exhibits anti-ulcer activity possibly by inhibition of Helicobacter.
Ikarugamycin (CAS 36531-78-9) References
- Human immunodeficiency virus type 1 Nef-induced CD4 cell surface downregulation is inhibited by ikarugamycin. | Luo, T., et al. 2001. J Virol. 75: 2488-92. PMID: 11160755
- Ripromycin and other polycyclic macrolactams from Streptomyces sp. Tü 6239: taxonomy, fermentation, isolation and biological properties. | Bertasso, M., et al. 2003. J Antibiot (Tokyo). 56: 364-71. PMID: 12817810
- Inhibition of the uptake of oxidized low-density lipoprotein in macrophage J774 by the antibiotic ikarugamycin. | Hasumi, K., et al. 1992. Eur J Biochem. 205: 841-6. PMID: 1572375
- Distinct endocytic pathways identified in tobacco pollen tubes using charged nanogold. | Moscatelli, A., et al. 2007. J Cell Sci. 120: 3804-19. PMID: 17940063
- Ikarugamycin induces DNA damage, intracellular calcium increase, p38 MAP kinase activation and apoptosis in HL-60 human promyelocytic leukemia cells. | Popescu, R., et al. 2011. Mutat Res. 709-710: 60-6. PMID: 21392513
- Heterologous reconstitution of ikarugamycin biosynthesis in E. coli. | Antosch, J., et al. 2014. Angew Chem Int Ed Engl. 53: 3011-4. PMID: 24519911
- Butremycin, the 3-hydroxyl derivative of ikarugamycin and a protonated aromatic tautomer of 5'-methylthioinosine from a Ghanaian Micromonospora sp. K310. | Kyeremeh, K., et al. 2014. Mar Drugs. 12: 999-1012. PMID: 24534843
- Mechanistic insights into polycycle formation by reductive cyclization in ikarugamycin biosynthesis. | Zhang, G., et al. 2014. Angew Chem Int Ed Engl. 53: 4840-4. PMID: 24706593
- New ikarugamycin derivatives with antifungal and antibacterial properties from Streptomyces zhaozhouensis. | Lacret, R., et al. 2014. Mar Drugs. 13: 128-40. PMID: 25551780
- Ikarugamycin: A Natural Product Inhibitor of Clathrin-Mediated Endocytosis. | Elkin, SR., et al. 2016. Traffic. 17: 1139-49. PMID: 27392092
- Biocatalytic Total Synthesis of Ikarugamycin. | Greunke, C., et al. 2017. Angew Chem Int Ed Engl. 56: 4351-4355. PMID: 28128495
- Ikarugamycin inhibits pancreatic cancer cell glycolysis by targeting hexokinase 2. | Jiang, SH., et al. 2020. FASEB J. 34: 3943-3955. PMID: 31944405
- A potent endocytosis inhibitor Ikarugamycin up-regulates TNF production. | Minamidate, A., et al. 2021. Biochem Biophys Rep. 27: 101065. PMID: 34286109
- Antibacterial Activity of Ikarugamycin against Intracellular Staphylococcus aureus in Bovine Mammary Epithelial Cells In Vitro Infection Model. | Saeed, SI., et al. 2021. Biology (Basel). 10: PMID: 34681057
- Polycyclic Tetramate Macrolactams-A Group of Natural Bioactive Metallophores. | Ding, L., et al. 2021. Front Chem. 9: 772858. PMID: 34869220
Inhibitor of:
γ1-Adaptin, γ2-Adaptin, ANKFY1, AP-2μ1, CCDC93, Clathrin LCB, Deltex (DTX; E3 ubiquitin ligase), E430025E21Rik, EG666955, EHD (EH Domain Containing), ENDOD1, Eps15L1, Rabenosyn-5, UNC93A, UNQ830, and ZNF408.Activator of:
FCHO2, NECAP 1, and Unc18-2.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ikarugamycin, 500 µg | sc-202179 | 500 µg | $189.00 |
