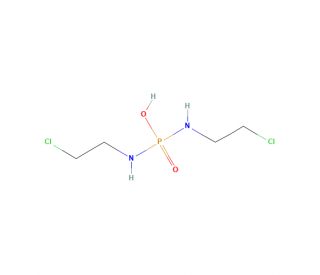
Ifosfamide mustard (CAS 31645-39-3)
QUICK LINKS
Ifosfamide mustard also know as Palifosfamide or 4-chloro-N-[4-[[[(2-chloroethyl)amino]carbonyl]amino]phenyl]benzamide, has been identified to have unique biological and pharmacological properties and has been examined for its potential to interact with various kinds of cancer cells. The synthesis of Ifosfamide mustard is a three-step process, which involves the reaction of 4-chlorobenzamide with 2-chloroethyl isocyanate, subsequently reacted with 4-amino-benzoyl chloride, and finally with 2-chloroethyl amine. The action mechanism of Ifosfamide mustard is not entirely clear at this point. It is speculated to function by hindering the activity of enzymes involved in DNA, RNA, and protein synthesis. This inhibition leads to the demise of cancer cells.
Ifosfamide mustard (CAS 31645-39-3) References
- Mechanistic aspects of the cytotoxic activity of glufosfamide, a new tumour therapeutic agent. | Seker, H., et al. 2000. Br J Cancer. 82: 629-34. PMID: 10682676
- Ifosfamide metabolites CAA, 4-OH-Ifo and Ifo-mustard reduce apical phosphate transport by changing NaPi-IIa in OK cells. | Patzer, L., et al. 2006. Kidney Int. 70: 1725-34. PMID: 17003823
- Variations in schedules of ifosfamide administration: a better understanding of its implications on pharmacokinetics through a randomized cross-over study. | Brain, EG., et al. 2007. Cancer Chemother Pharmacol. 60: 375-81. PMID: 17106751
- The effect of N-acetylcysteine on the antitumor activity of ifosfamide. | Chen, N., et al. 2011. Can J Physiol Pharmacol. 89: 335-43. PMID: 21609276
- Modulation of the control of mutagenic metabolites derived from cyclophosphamide and ifosfamide by stimulation of protein kinase A. | Oesch-Bartlomowicz, B., et al. 1990. Mutat Res. 232: 305-12. PMID: 2170835
- Genetically engineered V79 Chinese hamster cells metabolically activate the cytostatic drugs cyclophosphamide and ifosfamide. | Doehmer, J., et al. 1990. Environ Health Perspect. 88: 63-5. PMID: 2272335
- Synthesis and antitumor activity of two ifosfamide analogs with a five-membered ring. | Kutscher, B., et al. 1995. Arzneimittelforschung. 45: 323-6. PMID: 7741794
- D-19575--a sugar-linked isophosphoramide mustard derivative exploiting transmembrane glucose transport. | Pohl, J., et al. 1995. Cancer Chemother Pharmacol. 35: 364-70. PMID: 7850916
- Glutathione conjugation of the cytostatic drug ifosfamide and the role of human glutathione S-transferases. | Dirven, HA., et al. 1995. Chem Res Toxicol. 8: 979-86. PMID: 8555414
- Partitioning of ifosfamide and its metabolites between red blood cells and plasma. | Momerency, G., et al. 1996. J Pharm Sci. 85: 262-5. PMID: 8699325
- In vitro studies of the hyperthermic enhancement of activated ifosfamide (4-hydroperoxy-ifosfamide) and glucose isophosphoramide mustard. | Kutz, ME., et al. 1997. Cancer Chemother Pharmacol. 40: 167-71. PMID: 9182839
- Quantitation of suspensions (MESED). Application of MESED-gC/MS in the quantitation of ifosfamide mustard in erythrocytes, plasma, and plasma water | H. Dumez, G. Guetens, G. De Boeck, M. Highley, U. R. Tjaden, R. Maes, A. Hanauske, A. T. Van Oosterom, E. A. de Bruijn. 2001. Journal of Separation Science. Volume24, Issue2: 123-128.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ifosfamide mustard, 10 mg | sc-506140 | 10 mg | $408.00 |
