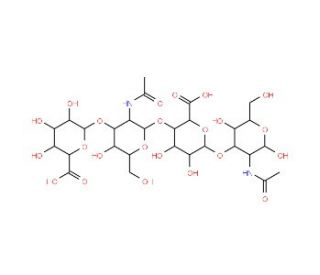
Hyaluronate Tetrasaccharide (CAS 57282-61-8)
QUICK LINKS
Hyaluronate tetrasaccharide, a truncated form of hyaluronic acid, has garnered significant interest in scientific research due to its role as a molecular probe and its potential applications in various fields. This tetrasaccharide consists of four sugar units derived from hyaluronic acid, a naturally occurring polysaccharide found in the extracellular matrix of connective tissues. In research, hyaluronate tetrasaccharide is primarily utilized as a molecular tool to investigate the interactions between hyaluronic acid and its binding partners, such as hyaluronidases, receptors, and other biomolecules. By studying the binding affinity and specificity of hyaluronate tetrasaccharide with these molecules, researchers gain insights into the structural and functional properties of hyaluronic acid and its biological roles in various physiological processes, including cell adhesion, migration, and inflammation. Furthermore, hyaluronate tetrasaccharide serves as a valuable substrate for enzymatic assays to characterize the activity of hyaluronidases and other hyaluronic acid-degrading enzymes. Additionally, this chemical entity has been explored as a potential agent for modulating hyaluronic acid metabolism in disease conditions, such as cancer and inflammatory disorders, by targeting specific molecular interactions involved in pathological processes. Overall, hyaluronate tetrasaccharide represents a versatile tool in scientific research for elucidating the biological functions of hyaluronic acid and developing novel strategies for disease intervention and tissue engineering applications.
Hyaluronate Tetrasaccharide (CAS 57282-61-8) References
- Hyaluronate tetrasaccharide- Cu(II) interaction: a NMR study. | D'Auria, G., et al. 2003. Biopolymers. 70: 260-9. PMID: 14517914
- Identification of the reaction products of the purified hyaluronidase from stonefish (Synanceja horrida) venom. | Sugahara, K., et al. 1992. Biochem J. 283 (Pt 1): 99-104. PMID: 1567384
- Effect of ionic strength and pH on the properties of purified bovine testicular hyaluronidase. | Gorham, SD., et al. 1975. Connect Tissue Res. 3: 17-25. PMID: 240644
- Concerted action of beta-glucuronidase and beta-acetylglucosaminidase on hyaluronodextrins. | Weissman, B., et al. 1975. Connect Tissue Res. 3: 7-15. PMID: 240646
- The cell surface hyaluronate binding sites of invasive human bladder carcinoma cells. | Nemec, RE., et al. 1987. Biochem Biophys Res Commun. 149: 249-57. PMID: 2446619
- Secondary structure of chondroitin sulphate in dimethyl sulphoxide. | Scott, JE., et al. 1983. Eur J Biochem. 130: 491-5. PMID: 6402367
- Secondary structure of hyaluronate in solution. A 1H-n.m.r. investigation at 300 and 500 MHz in [2H6]dimethyl sulphoxide solution. | Scott, JE., et al. 1984. Biochem J. 220: 197-205. PMID: 6743260
- Secondary structures of hyaluronate and chondroitin sulphates. A 1H n.m.r. study of NH signals in dimethyl sulphoxide solution. | Scott, JE., et al. 1981. Biochem J. 199: 829-32. PMID: 6803773
- Substrate specificity and regulation of activity of rat liver beta-D-glucuronidase. | Niemann, R. and Buddecke, E. 1982. Hoppe Seylers Z Physiol Chem. 363: 591-8. PMID: 7106706
- Measurements of reducing end groups on bovine vitreous-humour hyaluronic acid by reaction with [14C]cyanide. | Swann, DA., et al. 1982. Biochem J. 207: 409-14. PMID: 7165701
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Hyaluronate Tetrasaccharide, 1 mg | sc-295164 | 1 mg | $381.00 | |||
Hyaluronate Tetrasaccharide, 5 mg | sc-295164A | 5 mg | $1336.00 |
