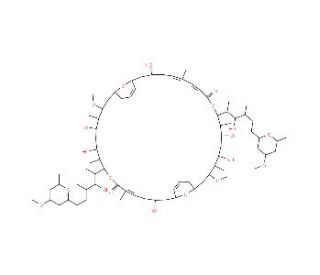
Hurghadolide A
QUICK LINKS
Hurghadolide A is a naturally occurring lactone derived from the Hurghada sp. sea sponge, notable for its complex molecular structure comprising a unique macrocyclic lactone ring. The chemical has piqued the interest of researchers primarily in the fields of organic and analytical chemistry due to its complex synthesis and intriguing biological activities. The mechanism by which Hurghadolide A operates involves interaction with biological membranes, attributed to its lipophilic lactone ring, which facilitates penetration into lipid-rich environments. This interaction is critical for studies on membrane dynamics and structural biology, where understanding the integration and effects of such compounds within lipid bilayers can reveal much about membrane-based processes and stability. In research, Hurghadolide A has been utilized as a probe to discern the behavior of small molecules within cellular membranes, offering insights that are crucial for the development of new materials and chemicals that mimic or disrupt natural biological functions. Furthermore, its unique structure serves as a model for synthetic chemists aiming to design and synthesize novel compounds with similar or enhanced properties, thus driving advancements in the synthesis of macrocyclic compounds which are notoriously challenging due to their size and complexity.
Hurghadolide A References
- Hurghadolide A and swinholide I, potent actin-microfilament disrupters from the Red Sea sponge Theonella swinhoei. | Youssef, DT. and Mooberry, SL. 2006. J Nat Prod. 69: 154-7. PMID: 16441091
- Natural products from the Lithistida: a review of the literature since 2000. | Winder, PL., et al. 2011. Mar Drugs. 9: 2643-2682. PMID: 22363244
- Efficacy of some natural compounds as antifungal agents. | Vengurlekar, S., et al. 2012. Pharmacogn Rev. 6: 91-9. PMID: 23055634
- The Swinholide Biosynthesis Gene Cluster from a Terrestrial Cyanobacterium, Nostoc sp. Strain UHCC 0450. | Humisto, A., et al. 2018. Appl Environ Microbiol. 84: PMID: 29150506
- Natural Products Repertoire of the Red Sea. | El-Hossary, EM., et al. 2020. Mar Drugs. 18: PMID: 32899763
- Marine-Derived Macrolides 1990-2020: An Overview of Chemical and Biological Diversity. | Zhang, H., et al. 2021. Mar Drugs. 19: PMID: 33806230
- The Significant Role of the Microfilament System in Tumors. | Jiang, X., et al. 2021. Front Oncol. 11: 620390. PMID: 33816252
- Bioactive Compounds from Marine Sponges: Fundamentals and Applications. | Varijakzhan, D., et al. 2021. Mar Drugs. 19: PMID: 33925365
- Deep-Sea Sponges and Corals off the Western Coast of Florida-Intracellular Mechanisms of Action of Bioactive Compounds and Technological Advances Supporting the Drug Discovery Pipeline. | Iskandar, M., et al. 2023. Mar Drugs. 21: PMID: 38132936
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Hurghadolide A, 10 µg | sc-221732 | 10 µg | $265.00 |
