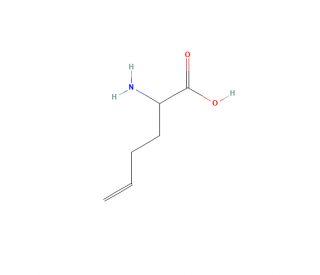
Homoallylglycine (CAS 16258-05-2)
QUICK LINKS
Homoallylglycine functions as an inhibitor of glutamate decarboxylase, the enzyme responsible for the conversion of glutamate to GABA. By inhibiting this enzyme, homoallylglycine disrupts the synthesis of GABA, an important neurotransmitter in the central nervous system. This disruption can be used to study the role of GABA in various physiological and pathological processes. Homoallylglycine can be utilized to investigate the impact of altered GABA levels on neuronal excitability and synaptic transmission. Its mechanism of action involves binding to the active site of glutamate decarboxylase, thereby preventing the conversion of glutamate to GABA. This inhibition can provide insights into the regulatory mechanisms of GABA synthesis and its implications for neurological function.
Homoallylglycine (CAS 16258-05-2) References
- Attenuation of the editing activity of the Escherichia coli leucyl-tRNA synthetase allows incorporation of novel amino acids into proteins in vivo. | Tang, Y. and Tirrell, DA. 2002. Biochemistry. 41: 10635-45. PMID: 12186549
- Conformationally restricted analogs of deoxynegamycin. | Raju, B., et al. 2004. Bioorg Med Chem Lett. 14: 3103-7. PMID: 15149653
- Covalent capture: a natural complement to self-assembly. | Hartgerink, JD. 2004. Curr Opin Chem Biol. 8: 604-9. PMID: 15556403
- Parallel synthesis and biological evaluation of different sizes of bicyclo[2,3]-Leu-enkephalin analogues. | Gu, X., et al. 2005. Biopolymers. 80: 151-63. PMID: 15660379
- Olefin metathesis for site-selective protein modification. | Lin, YA., et al. 2009. Chembiochem. 10: 959-69. PMID: 19343741
- A metabolic alkene reporter for spatiotemporally controlled imaging of newly synthesized proteins in Mammalian cells. | Song, W., et al. 2010. ACS Chem Biol. 5: 875-85. PMID: 20666508
- A biosynthetic route to photoclick chemistry on proteins. | Wang, J., et al. 2010. J Am Chem Soc. 132: 14812-8. PMID: 20919707
- Photoinducible bioorthogonal chemistry: a spatiotemporally controllable tool to visualize and perturb proteins in live cells. | Lim, RK. and Lin, Q. 2011. Acc Chem Res. 44: 828-39. PMID: 21609129
- Incorporation of Methionine Analogues Into Bombyx mori Silk Fibroin for Click Modifications. | Teramoto, H. and Kojima, K. 2015. Macromol Biosci. 15: 719-27. PMID: 25644632
- Conformation-Enabled Total Syntheses of Ohmyungsamycins A and B and Structural Revision of Ohmyungsamycin B. | Hur, J., et al. 2018. Angew Chem Int Ed Engl. 57: 3069-3073. PMID: 29380472
- Modification of Proteins Using Olefin Metathesis. | Messina, MS. and Maynard, HD. 2020. Mater Chem Front. 4: 1040-1051. PMID: 34457313
- Probing Site-Selective Conjugation Chemistries for the Construction of Homogeneous Synthetic Glycodendriproteins. | Cobo, I., et al. 2022. Chembiochem. 23: e202200020. PMID: 35322922
- The silica mineralisation properties of synthetic Silaffin-1A1 (synSil-1A1). | Daus, F., et al. 2022. Org Biomol Chem. 20: 3387-3396. PMID: 35362502
- Efficient introduction of alkene functionality into proteins in vivo. | van Hest, JC. and Tirrell, DA. 1998. FEBS Lett. 428: 68-70. PMID: 9645477
- Large scale enantiomeric synthesis, purification, and characterization of ω-unsaturated amino acids via a Gly-Ni(II)-BPB-complex | Xuyuan Gu, John M. Ndungu, Wei Qiu †, Jinfa Ying, Michael D. Carducci, Hank Wooden, Victor J. Hruby. 2004. Tetrahedron. 60: 8233-8243.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Homoallylglycine, 50 mg | sc-490295 | 50 mg | $367.00 |
