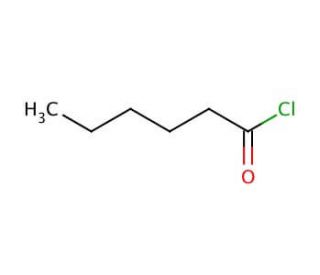
Hexanoyl chloride (CAS 142-61-0)
ENLACES RÁPIDOS
Hexanoyl chloride, o n-hexanoyl chloride, es un compuesto orgánico. Este líquido incoloro posee volatilidad y emite un olor penetrante. Sus aplicaciones abarcan diversos ámbitos, incluyendo la síntesis orgánica y la producción de polímeros. Hexanoyl chloride sirve como un reactivo crucial en la investigación científica, contribuyendo a la síntesis de diversos compuestos orgánicos, como aminoácidos, péptidos y carbohidratos. En el ámbito de la investigación científica, el hexanoyl chloride actúa como un reactivo, participando en la síntesis orgánica. A través de sus interacciones con alcoholes, aminas y otros compuestos orgánicos, facilita la formación de ésteres, amidas y diversos otros compuestos. Su naturaleza versátil se extiende a la preparación de polímeros y otros materiales, mejorando su síntesis y funcionalidad. Como resultado, el hexanoyl chloride permite la exploración de numerosas vías de investigación y el desarrollo de compuestos y materiales innovadores.
Hexanoyl chloride (CAS 142-61-0) Referencias
- Síntesis de nuevos tensioactivos a base de azúcares y evaluación de sus actividades hemolíticas. | Neimert-Andersson, K., et al. 2006. J Org Chem. 71: 3623-6. PMID: 16626152
- Agregados de piromelitamida y su respuesta a estímulos aniónicos. | Webb, JE., et al. 2007. J Am Chem Soc. 129: 7155-62. PMID: 17497782
- Nanopartículas magnéticas estabilizadas con N-hexanoil quitosano: potenciación de la expresión génica mediada por adenovirus tanto in vitro como in vivo. | Bhattarai, SR., et al. 2008. Nanomedicine. 4: 146-54. PMID: 18374634
- Síntesis total y cribado antibacteriano de (±)-7-butil-6,8-dihidroxi-3-pentil-3,4-dihidroisocromen-1-ona. | Saeed, A., et al. 2013. J Asian Nat Prod Res. 15: 1112-22. PMID: 23869649
- Síntesis fácil de isotiocianatos de acil quitosano y su aplicación a derivados de quitosano apendicados con porfirina. | Shibano, M., et al. 2014. Carbohydr Polym. 113: 279-85. PMID: 25256486
- Películas de celulosa hidrófoba con excelente resistencia y tenacidad mediante acilación activada por molienda de bolas de celulosa microfibrillada. | Deng, S., et al. 2016. Carbohydr Polym. 154: 129-38. PMID: 27577904
- Una Visión Complementaria y Revisada de la N-Acilación del Quitosano con Cloruro de Hexanoilo. | Reis, B., et al. 2021. Mar Drugs. 19: PMID: 34356810
- Síntesis de hexanoato de quitina termoplástico-injerto-poli(ε-caprolactona). | Nakashima, A., et al. 2022. Carbohydr Polym. 280: 119024. PMID: 35027126
- Esterificación de la lignina con ácidos grasos de cadena larga para la estabilización de emulsiones Pickering de aceite en agua. | Shorey, R. and Mekonnen, TH. 2023. Int J Biol Macromol. 230: 123143. PMID: 36641016
Información sobre pedidos
| Nombre del producto | Número de catálogo | UNIDAD | Precio | CANTIDAD | Favoritos | |
Hexanoyl chloride, 25 ml | sc-250109 | 25 ml | $45.00 | |||
Hexanoyl chloride, 100 ml | sc-250109A | 100 ml | $67.00 |
