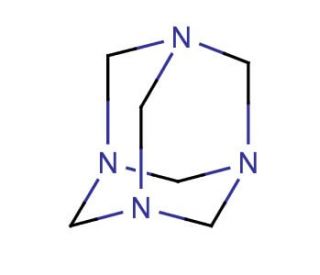
Hexamethylenetetramine (CAS 100-97-0)
QUICK LINKS
Hexamethylenetetramine, commonly known as hexamine or methenamine, serves as a vital chemical compound in various research applications owing to its unique properties and versatile functionalities. One of the key mechanisms of action of hexamethylenetetramine is its role as a cross-linking agent in the synthesis of organic polymers and resins. This compound undergoes hydrolysis in the presence of water and acidic conditions to release formaldehyde, which subsequently reacts with amino groups to form methylene bridges, leading to the cross-linking of polymer chains. This cross-linking capability finds extensive use in the development of adhesives, coatings, and composite materials with enhanced mechanical strength, thermal stability, and chemical resistance. Furthermore, hexamethylenetetramine is employed as a precursor in the synthesis of metal-organic frameworks (MOFs) and coordination polymers due to its ability to coordinate with metal ions and form stable complexes. These MOFs exhibit a wide range of applications in gas storage, catalysis, sensing, and drug delivery systems, contributing significantly to materials science and nanotechnology research. Additionally, hexamethylenetetramine serves as a nitrogen source in the preparation of nitrogen-containing compounds, such as pharmaceuticals, dyes, and explosives, facilitating advancements in organic synthesis and chemical engineering. Overall, the diverse mechanisms and research applications of hexamethylenetetramine underscore its importance as a fundamental building block in various scientific disciplines, including polymer chemistry, materials science, and nanotechnology.
Hexamethylenetetramine (CAS 100-97-0) References
- [In vitro fungistatic effects of formaldehyde, hexamethylenetetramine & related compounds in various hydrogen ion concentrations. I. Data on compounds with fungistatic properties]. | WEUFFEN, W. and POHLOUDEK-FABINI, R. 1959. Arzneimittelforschung. 9: 365-8. PMID: 13670866
- Epitaxial growth of the zinc oxide nanorods, their characterization and in vitro biocompatibility studies. | Gopikrishnan, R., et al. 2011. J Mater Sci Mater Med. 22: 2301-9. PMID: 21823031
- Synthesis, characterisation and in vitro cytotoxicity of mixed ligand Pt(ii) oxadiazoline complexes with hexamethylenetetramine and 7-nitro-1,3,5-triazaadamantane. | Sieste, S., et al. 2017. Dalton Trans. 46: 12226-12238. PMID: 28875218
- Improved corrosion resistance of commercially pure magnesium after its modification by plasma electrolytic oxidation with organic additives. | Echeverry-Rendon, M., et al. 2018. J Biomater Appl. 33: 725-740. PMID: 30444445
- A Study of the Chemistries, Growth Mechanisms, and Antibacterial Properties of Cerium- and Yttrium-Containing Nanoparticles. | Bassous, NJ., et al. 2021. ACS Biomater Sci Eng. 7: 1787-1807. PMID: 33966381
- Development of an LC-MS/MS Assay and Toxicokinetic Characterization of Hexamethylenetetramine in Rats. | Kim, W., et al. 2023. Toxics. 11: PMID: 37112564
- Synthesis and Application of Carbon Quantum Dots Derived from Carbon Black in Bioimaging. | Molaei, MJ. 2024. J Fluoresc. 34: 213-226. PMID: 37191828
- Prophylactic and antidotal effects of hexamethylenetetramine against phosgene poisoning in rabbits. | Frosolono, MF. 1985. Toxicol Ind Health. 1: 101-16. PMID: 3842184
- Non-carcinogenicity of hexamethylenetetramine in mice and rats. | Della Porta, G., et al. 1968. Food Cosmet Toxicol. 6: 707-15. PMID: 4308809
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Hexamethylenetetramine, 5 g | sc-211588 | 5 g | $36.00 | |||
Hexamethylenetetramine, 250 g | sc-211588A | 250 g | $82.00 |
