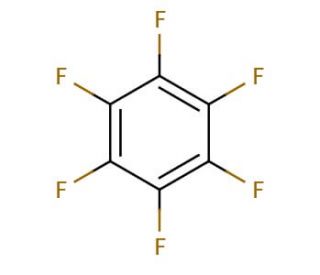
Hexafluorobenzene (CAS 392-56-3)
QUICK LINKS
Hexafluorobenzene (HFB) is a perfluorinated compound, possessing diverse applications in chemistry, biochemistry, and material science. It is a clear, volatile liquid with a distinct, pleasant scent. Acting as a intermediary, Hexafluorobenzene aids in the production of various organic compounds like dyes and pigments. Additionally, it serves as a key component in the synthesis of polymers, surfactants, and other materials. In the realm of scientific research, Hexafluorobenzene finds wide-ranging utility as a reagent in organic synthesis, a solvent for chemical reactions, and a starting point for the creation of fluorinated compounds. Furthermore, it plays a significant role in the generation of organofluorine compounds, including fluorinated alcohols and ethers. For reactions. eg. Grignards: J.Organomet.
Hexafluorobenzene (CAS 392-56-3) References
- Hexafluorobenzene photochemistry: wellspring of fluorocarbon structures. | Lemal, DM. 2001. Acc Chem Res. 34: 662-71. PMID: 11513574
- Hexafluorobenzene acts in the spinal cord, whereas o-difluorobenzene acts in both brain and spinal cord, to produce immobility. | Antognini, JF., et al. 2007. Anesth Analg. 104: 822-8. PMID: 17377088
- The two structures of the hexafluorobenzene radical cation C6F6(*+). | Shorafa, H., et al. 2009. Angew Chem Int Ed Engl. 48: 5845-7. PMID: 19582741
- Hexafluorobenzene: a powerful solvent for a noncovalent stereoselective organocatalytic Michael addition reaction. | Lattanzi, A., et al. 2012. Chem Commun (Camb). 48: 1650-2. PMID: 22190150
- Hexafluorobenzene in comparison with perfluoro-15-crown-5-ether for repeated monitoring of oxygenation using 19F MRI in a mouse model. | Mignion, L., et al. 2013. Magn Reson Med. 69: 248-54. PMID: 22442096
- Photophysics of fluorinated benzene. III. Hexafluorobenzene. | Mondal, T., et al. 2012. J Chem Phys. 137: 054311. PMID: 22894352
- Hexafluorobenzene under Extreme Conditions. | Pravica, M., et al. 2016. J Phys Chem B. 120: 2854-8. PMID: 26910443
- Synthesis of Fluorine-Doped Hydrophilic Carbon Nanoparticles from Hexafluorobenzene by Femtosecond Laser Pulses. | Okamoto, T., et al. 2017. Chemphyschem. 18: 1007-1011. PMID: 27557055
- High-Temperature Reactions of Hexafluorobenzene. | Antonucci, JM. and Wall, LA. 1966. J Res Natl Bur Stand A Phys Chem. 70A: 473-480. PMID: 31824014
- IR absorption spectra of hexafluorobenzene anions and pentafluorophenyl radicals in solid argon. | Chou, SL., et al. 2021. Spectrochim Acta A Mol Biomol Spectrosc. 252: 119524. PMID: 33582441
- Role of the Perfluoro Effect in the Selective Photochemical Isomerization of Hexafluorobenzene. | Cox, JM., et al. 2021. J Am Chem Soc. 143: 7002-7012. PMID: 33938749
- Temperature-Dependent Photoluminescence of Hexafluorobenzene-Intercalated Phenethylammonium Tin Iodide 2D Perovskite. | Dutta, T., et al. 2021. Chem Asian J. 16: 2745-2751. PMID: 34342155
- Hexafluorobenzene: a sensitive 19F NMR indicator of tumor oxygenation. | Mason, RP., et al. 1996. NMR Biomed. 9: 125-34. PMID: 8892399
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Hexafluorobenzene, 5 g | sc-257584 | 5 g | $39.00 | |||
Hexafluorobenzene, 25 g | sc-257584A | 25 g | $125.00 |
