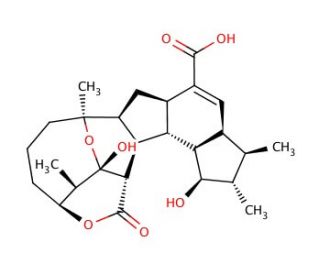
Hexacyclinic acid
QUICK LINKS
Hexacyclinic acid, a chemical compound that is part of a class of substances known as polyketides, is notable for its complex molecular structure and biological activity. While specific CAS registry numbers and detailed empirical data might sometimes be sparse for lesser-known compounds like this, hexacyclinic acid′s chemical profile suggests that it is derived from a polyketide synthase pathway, typical of many natural products with diverse bioactivities. Polyketides like hexacyclinic acid are produced by various microorganisms and plants and are characterized by their assembly from acetyl and propionyl coenzyme A units through a series of enzymatic steps. Research into hexacyclinic acid has focused on its biosynthesis and the unique enzymatic mechanisms involved in constructing its polyketide backbone. In academic and scientific studies, hexacyclinic acid has been of interest primarily for its potential role in inhibiting certain biological processes at the cellular level, likely through mechanisms involving the disruption of normal biochemical pathways. Its structure and activity have made it a candidate for studying enzyme-substrate interactions within polyketide synthases, offering insights into the synthesis of complex natural products and providing a framework for developing synthetic analogs with optimized properties. This research is crucial for advancing the understanding of polyketide biosynthesis and exploring novel applications in biotechnology.
Hexacyclinic acid References
- Hexacyclinic acid, a Polyketide from Streptomyces with a Novel Carbon Skeleton We express our thanks to Hoechst AG (Frankfurt) for providing us with Streptomyces cellulosae (strain S 1013) and Mr. M. Decke for excellent technical assistance. We wish to thank Prof. Dr. W. Beil for determining the cytotoxic activity. This work was supported by the Fonds der Chemischen Industrie. | Höfs, R., et al. 2000. Angew Chem Int Ed Engl. 39: 3258-3261. PMID: 11028069
- Intramolecular allenolate acylations in studies toward a synthesis of FR182877. | Vanderwal, CD., et al. 2001. Org Lett. 3: 4307-10. PMID: 11784204
- A cycloaddition cascade approach to the total synthesis of (-)-FR182877. | Evans, DA. and Starr, JT. 2003. J Am Chem Soc. 125: 13531-40. PMID: 14583050
- Synthesis of the A,B,C-ring system of hexacyclinic acid. | Stellfeld, T., et al. 2004. Org Lett. 6: 3889-92. PMID: 15496056
- Free-radical-5-exo-trig cyclization of chiral 3-silylhepta-1,6-dienes: concise approach to the A-B-C ring core of hexacyclinic acid. | James, P., et al. 2005. J Org Chem. 70: 7985-95. PMID: 16277319
- Tandem intramolecular Michael-aldol reaction as a tool for the construction of the C-ring of hexacyclinic acid. | Stelmakh, A., et al. 2006. Org Lett. 8: 3485-8. PMID: 16869641
- Studies on transannulation reactions across a nine-membered ring: the synthesis of natural product-like structures. | Iqbal, M., et al. 2011. Org Biomol Chem. 9: 5062-78. PMID: 21597625
- Synthesis of a model DEF-ring core of hexacyclinic acid. | Clarke and Paul A., et al. 2003. Chemical communications. 13: 1560-1561.
- An iodocyclisation/elimination approach to a DEF-ring core of FR182877. | Clarke, et al. 2004. Tetrahedron letters. 45.5: 927-929.
- ynthetic studies on the DEF-rings of FR182877 and hexacyclinic acid. | Clarke and Paul A., et al. 2005. Tetrahedron. 61.2: 353-363.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Hexacyclinic acid, 1 mg | sc-221718 | 1 mg | $317.00 | |||
Hexacyclinic acid, 5 mg | sc-221718A | 5 mg | $960.00 |
