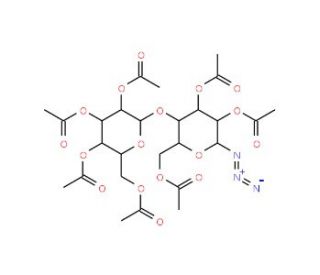
Hepta-O-acetyl-β-lactosyl azide (CAS 30854-62-7)
QUICK LINKS
Hepta-O-acetyl-β-lactosyl azide is a derivative of lactose, modified by acetylation to produce a glycosyl azide. In research, this compound is primarily utilized for its application in carbohydrate chemistry, where it serves as a key intermediate in glycosylation reactions. The azide functionality allows for efficient conjugation to various molecular scaffolds through click chemistry, specifically the Huisgen cycloaddition reaction with alkynes, enabling the formation of stable triazole linkages. This characteristic makes it a useful tool in creating glycoconjugates and glycosylated compounds that are relevant to studying the structural diversity of carbohydrates and their roles in biological recognition processes. Hepta-O-acetyl-β-lactosyl azide has been used in synthesizing glycoproteins, glycolipids, and oligosaccharides that mimic biologically significant carbohydrates. These synthetic constructs provide insights into carbohydrate-protein interactions, cellular recognition, and the mechanisms of pathogenic binding. Moreover, this compound is helpful in exploring the structural analysis of oligosaccharides via chemical tagging and as a building block in the development of carbohydrate-based vaccines and diagnostic tools. Its robust chemical reactivity and affinity for glycosylation pathways make it an invaluable resource for elucidating the complexities of carbohydrate chemistry in biochemical research.
Hepta-O-acetyl-β-lactosyl azide (CAS 30854-62-7) References
- Expedient synthesis of triazole-linked glycosyl amino acids and peptides. | Kuijpers, BH., et al. 2004. Org Lett. 6: 3123-6. PMID: 15330603
- Synthesis and gelation property of a series of disaccharide triazole derivatives. | Okafor, IS. and Wang, G. 2017. Carbohydr Res. 451: 81-94. PMID: 28987928
- Synthesis of Galectin Inhibitors by Regioselective 3'-O-Sulfation of Vanillin Lactosides Obtained under Phase Transfer Catalysis. | Belkhadem, K., et al. 2020. Molecules. 26: PMID: 33383774
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Hepta-O-acetyl-β-lactosyl azide, 100 mg | sc-300790 | 100 mg | $630.00 |
