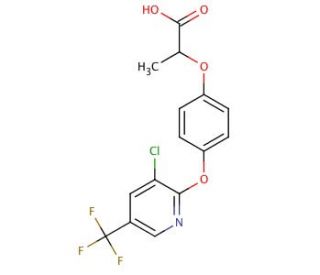
Haloxyfop (CAS 69806-34-4)
QUICK LINKS
Haloxyfop is a chemical compound that is primarily used in agricultural research focused on the study of herbicidal action, particularly in the control of grassy weeds. Researchers investigate the mechanisms by which haloxyfop acts as a post-emergence herbicide, often exploring its inhibition of fatty acid synthesis in target plant species. This inhibition leads to the disruption of cell membrane formation and plant growth. The compound′s selectivity and effectiveness in various crop systems are key areas of study, aiming to optimize weed management practices while minimizing impacts on non-target species. Additionally, research on haloxyfop includes environmental fate studies, where its persistence, degradation, and movement within ecosystems are monitored to ensure responsible usage and to inform regulatory decisions.
Haloxyfop (CAS 69806-34-4) References
- Molecular basis for the inhibition of the carboxyltransferase domain of acetyl-coenzyme-A carboxylase by haloxyfop and diclofop. | Zhang, H., et al. 2004. Proc Natl Acad Sci U S A. 101: 5910-5. PMID: 15079078
- Inhibition of Acetyl-CoA Carboxylase Activity by Haloxyfop and Tralkoxydim. | Secor, J. and Cséke, C. 1988. Plant Physiol. 86: 10-2. PMID: 16665846
- Haloxyfop Inhibition of the Pyruvate and the alpha-Ketoglutarate Dehydrogenase Complexes of Corn (Zea mays L.) and Soybean (Glycine max [L.] Merr.). | Cho, HY., et al. 1988. Plant Physiol. 87: 334-40. PMID: 16666143
- A different mechanism for the inhibition of the carboxyltransferase domain of acetyl-coenzyme A carboxylase by tepraloxydim. | Xiang, S., et al. 2009. Proc Natl Acad Sci U S A. 106: 20723-7. PMID: 19926852
- Trypanosoma brucei: inhibition of acetyl-CoA carboxylase by haloxyfop. | Vigueira, PA. and Paul, KS. 2012. Exp Parasitol. 130: 159-65. PMID: 22119241
- Allelic mutations in acetyl-coenzyme A carboxylase confer herbicide tolerance in maize. | Marshall, LC., et al. 1992. Theor Appl Genet. 83: 435-42. PMID: 24202589
- Stereoselective quantitation of haloxyfop in environment samples and enantioselective degradation in soils. | Sun, M., et al. 2015. Chemosphere. 119: 583-589. PMID: 25128890
- Inhibition of plant acetyl-coenzyme A carboxylase by the herbicides sethoxydim and haloxyfop. | Burton, JD., et al. 1987. Biochem Biophys Res Commun. 148: 1039-44. PMID: 2891354
- Unravelling the binding affinity between model transport protein and a prospective tuberculosis therapeutic agent: a spectroscopic and theoretical simulation exploration. | Wang, L., et al. 2019. J Biomol Struct Dyn. 37: 4507-4521. PMID: 30663540
- Molecular characteristics of the first case of haloxyfop-resistant Poa annua. | Ghanizadeh, H., et al. 2020. Sci Rep. 10: 4231. PMID: 32144361
- Haloxyfop determination by gas chromatography/tandem mass spectrometry in eggs. | Lehner, AF., et al. 2020. Rapid Commun Mass Spectrom. 34: e8895. PMID: 32662916
- Toxicity of the herbicides diuron, propazine, tebuthiuron, and haloxyfop to the diatom Chaetoceros muelleri. | Thomas, MC., et al. 2020. Sci Rep. 10: 19592. PMID: 33177549
- Species-dependent induction of peroxisome proliferation by haloxyfop, an aryloxyphenoxy herbicide. | Stott, WT., et al. 1995. Fundam Appl Toxicol. 28: 71-9. PMID: 8566486
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Haloxyfop, 100 mg | sc-235275 | 100 mg | $109.00 |
