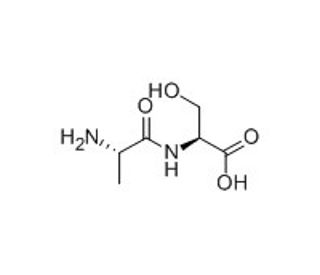
H-Ala-Ser-OH (CAS 3303-41-1)
QUICK LINKS
H-Ala-Ser-OH, a dipeptide composed of alanine and serine, serves as a valuable research chemical for understanding peptide synthesis and protease activity. This simple peptide is instrumental in studying the substrate specificity of various serine proteases and other enzyme classes that act on peptide bonds. Researchers utilize H-Ala-Ser-OH to investigate the fundamental aspects of peptide bond formation and degradation, which are critical in cellular processes such as protein turnover and signal transduction. The compound′s ability to act as a substrate for peptide synthesizing enzymes also provides insights into the mechanisms of enzyme catalysis, particularly how molecular structure affects the rate and specificity of enzymatic reactions. In biochemical studies, H-Ala-Ser-OH is used to calibrate and optimize conditions for peptide synthesis and for assays measuring enzyme activity. This dipeptide helps delineate how alterations in peptide chain length and composition affect the activity of enzymes that are crucial for maintaining cellular function and integrity. By contributing to a deeper understanding of peptide behavior and enzyme interaction, H-Ala-Ser-OH aids in the broader exploration of biochemical pathways involving peptides.
H-Ala-Ser-OH (CAS 3303-41-1) References
- Affinity and translocation relationships via hPEPT1 of H-X aa-Ser-OH dipeptides: evaluation of H-Phe-Ser-OH as a pro-moiety for ibuprofen and benzoic acid prodrugs. | Omkvist, DH., et al. 2011. Eur J Pharm Biopharm. 77: 327-31. PMID: 21147219
- Kinetic studies on the alkali-catalyzed hydrolysis and epimerization of model alkyl and hydroxyalkyl di- and tripeptides. | Noll, BW., et al. 1974. Biochemistry. 13: 5164-9. PMID: 4433513
- Studies with model substances on the mechanism of the formic acid-induced reversible inactivation of protein enzymes. | Josefsson, L. 1966. Biochim Biophys Acta. 115: 148-59. PMID: 5936231
- Proton NMR, EPR and absorption spectra studies of palladium (II) and copper (II) complexes with L-alanyl-L-serine | Kozłowski, H. and Siatecki, Z. 1978. Chemical Physics Letters. 54(3): 498-501.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
H-Ala-Ser-OH, 250 mg | sc-295038 | 250 mg | $218.00 | |||
H-Ala-Ser-OH, 1 g | sc-295038A | 1 g | $667.00 |
