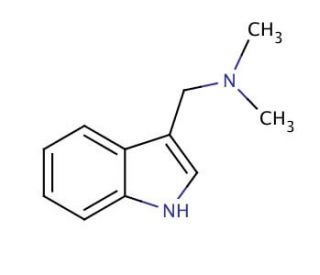
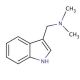
Gramine (CAS 87-52-5)
QUICK LINKS
Gramine functions as a plant alkaloid. It acts as a growth inhibitor, affecting the development of plant cells and tissues. Gramine′s mechanism of action involves interfering with the synthesis of proteins and nucleic acids in plants, ultimately leading to growth suppression. At the molecular level, it disrupts the function of ribosomes, which are useful for protein synthesis, and inhibits the activity of certain enzymes involved in nucleic acid production. This disruption at the cellular level results in stunted growth and reduced biomass accumulation in plants. Gramine′s role in experimental applications involves studying its effects on plant growth and development, as well as its potential applications in agricultural study.
Gramine (CAS 87-52-5) References
- Differential Impact of Plant Secondary Metabolites on the Soil Microbiota. | Schütz, V., et al. 2021. Front Microbiol. 12: 666010. PMID: 34122379
- Synthesis, antioxidant and cytoprotective activity evaluation of C-3 substituted indole derivatives. | Jasiewicz, B., et al. 2021. Sci Rep. 11: 15425. PMID: 34326403
- Gramine promotes functional recovery after spinal cord injury via ameliorating microglia activation. | Lu, X., et al. 2021. J Cell Mol Med. 25: 7980-7992. PMID: 34382745
- 3-Indoleacetonitrile Is Highly Effective in Treating Influenza A Virus Infection In Vitro and In Vivo. | Zhao, X., et al. 2021. Viruses. 13: PMID: 34452298
- Applications of the indole-alkaloid gramine modulate the assembly of individual members of the barley rhizosphere microbiota. | Maver, M., et al. 2021. PeerJ. 9: e12498. PMID: 34900424
- UPLC-Q-TOF-MS/MS analysis on the chemical composition of malts under different germination cycles and prepared with different processing methods. | Zou, J., et al. 2023. Fitoterapia. 165: 105313. PMID: 36179899
- OpenFL: the open federated learning library. | Foley, P., et al. 2022. Phys Med Biol. 67: PMID: 36198326
- Cytochrome P450s genes CYP321A9 and CYP9A58 contribute to host plant adaptation in the fall armyworm Spodoptera frugiperda. | He, L., et al. 2023. Pest Manag Sci. 79: 1783-1790. PMID: 36627818
- Impediment of Cancer by Dietary Plant-derived Alkaloids Through Oxidative Stress: Implications of PI3K/AKT Pathway in Apoptosis, Autophagy, and Ferroptosis. | Manogaran, P., et al. 2023. Curr Top Med Chem. 23: 860-877. PMID: 36635931
- Copper-Catalyzed C2- or C3-Thioglycosylation of Indoles with N-(Thioglycosides)succinimides: An Effective Strategy for the Total Synthesis of Isatindigotindolosides. | Huang, J., et al. 2023. Org Lett. 25: 528-532. PMID: 36646633
- Development of an HPLC-MS/MS Method for the Determination of Alkaloids in Lupins. | Eugelio, F., et al. 2023. Molecules. 28: PMID: 36838519
- Detection of a novel intramolecular rearrangement during gramine biosynthesis in barley using stable isotope-labeled tryptophan. | Ishikawa, E., et al. 2023. Biochem Biophys Rep. 34: 101439. PMID: 36843643
- Gramine protects against pressure overload-induced pathological cardiac hypertrophy through Runx1-TGFBR1 signaling. | Xu, L., et al. 2023. Phytomedicine. 114: 154779. PMID: 37023527
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Gramine, 25 g | sc-255191 | 25 g | $30.00 |
