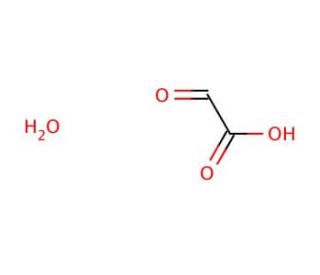
Glyoxylic acid monohydrate (CAS 563-96-2)
QUICK LINKS
Glyoxylic acid monohydrate is extensively researched in the fields of organic synthesis and biochemistry, particularly for its role as a building block in the synthesis of various organic compounds. Its reactivity is studied for the production of bioactive compounds, flavorings, and aroma compounds, where it acts as a key intermediate. Research on Glyoxylic acid monohydrate includes exploring its effectiveness in the formation of heterocyclic compounds and its use in the synthesis of vanillin, a widely used flavoring agent. The stability and handling of Glyoxylic acid monohydrate under various conditions are also significant areas of study, aiming to optimize its use in industrial and laboratory applications. Moreover, studies focus on the environmental impact of Glyoxylic acid monohydrate, particularly its biodegradability and potential effects on aquatic systems.
Glyoxylic acid monohydrate (CAS 563-96-2) References
- Simultaneous fluorescence histochemical demonstration of catecholamines and tryptophyl-peptides in endocrine cells. | Partanen, S. 1975. Histochemistry. 43: 295-303. PMID: 1184412
- A two-stage iterative process for the synthesis of poly-oxazoles. | Atkins, JM. and Vedejs, E. 2005. Org Lett. 7: 3351-4. PMID: 16018658
- Oxidative decarboxylative synthesis of 2-H-imidazolines from glyoxylic acid and 1,2-diamines. | Murai, K., et al. 2008. Chem Commun (Camb). 4498-500. PMID: 18802603
- Inhibiting inflammation and modulating oxidative stress in oxalate-induced nephrolithiasis with the Nrf2 activator dimethyl fumarate. | Zhu, J., et al. 2019. Free Radic Biol Med. 134: 9-22. PMID: 30599261
- One-Step Synthesis of Isoindolo[2,1-a]indol-6-ones via Tandem Pd-Catalyzed Aminocarbonylation and C-H Activation. | Čarný, T., et al. 2019. J Org Chem. 84: 12499-12507. PMID: 31507186
- Resveratrol Attenuates Oxalate-Induced Renal Oxidative Injury and Calcium Oxalate Crystal Deposition by Regulating TFEB-Induced Autophagy Pathway. | Wu, Y., et al. 2021. Front Cell Dev Biol. 9: 638759. PMID: 33718378
- Sequential Continuous-Flow Synthesis of 3-Aryl Benzofuranones. | Xin, HL., et al. 2021. Chem Asian J. 16: 1906-1910. PMID: 34137489
- Porous three-dimensional network of Pd-Cu aerogel toward formic acid oxidation. | Douk, AS., et al. 2018. RSC Adv. 8: 23539-23545. PMID: 35540256
- Biochemical analysis of catecholamines in small intensely fluorescent (SIF) cell clusters of the rat superior cervical ganglion. | Gerold, N., et al. 1982. J Neurosci Methods. 6: 287-92. PMID: 7144241
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Glyoxylic acid monohydrate, 10 g | sc-250065 | 10 g | $27.00 | |||
Glyoxylic acid monohydrate, 25 g | sc-250065A | 25 g | $37.00 |
