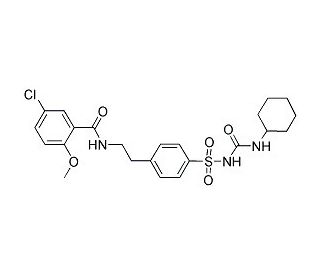
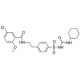
Glyburide (Glibenclamide) (CAS 10238-21-8)
See product citations (36)
QUICK LINKS
Glyburide, also known as Glibenclamide, is a sulfonylurea compound that regulates insulin production by modulating ATP-dependent K+ channels. These channels, found in the beta cells of the pancreas, are depolarized by sulfonylureas, leading to the release of Ca2+ and subsequent stimulation of insulin production, thereby improving glucose control. By binding to specific receptors on the surface of pancreatic beta cells, Glyburide (Glibenclamide) triggers the release of insulin. Additionally, Glyburide (Glibenclamide) acts as a blocker for ATP-dependent K+ channels (KIR6, KATP) and CFTR Cl- channels. Extensive experiments have been conducted to explore the impact of Glyburide (Glibenclamide) on glucose metabolism, insulin sensitivity, lipid metabolism, cardiovascular function, and various physiological and biochemical parameters. In vitro studies have investigated Glyburide (Glibenclamide)′s mechanism of action, effects on cell proliferation and differentiation, insulin secretion, as well as its influence on the expression of genes and proteins associated with glucose metabolism and other metabolic pathways.
Glyburide (Glibenclamide) (CAS 10238-21-8) References
- Effects of glyburide (glibenclamide) on myocardial function in Langendorff perfused rabbit heart and on myocardial contractility and slow calcium current in guinea-pig single myocytes. | Khatib, SY. and Boyett, MR. 2003. Mol Cell Biochem. 242: 81-7. PMID: 12619869
- Hydroxyl-substituted sulfonylureas as potent inhibitors of specific [3H]glyburide binding to rat brain synaptosomes. | Hill, RA., et al. 2003. Bioorg Med Chem. 11: 2099-113. PMID: 12670661
- Glibenclamide, a blocker of K+(ATP) channels, shows antileishmanial activity in experimental murine cutaneous leishmaniasis. | Serrano-Martín, X., et al. 2006. Antimicrob Agents Chemother. 50: 4214-6. PMID: 17015627
- High affinity [3H]glibenclamide binding sites in rat neuronal and cardiac tissue: localization and developmental characteristics. | Miller, JA., et al. 1991. J Pharmacol Exp Ther. 256: 358-64. PMID: 1899120
- The receptor for antidiabetic sulfonylureas controls the activity of the ATP-modulated K+ channel in insulin-secreting cells. | Schmid-Antomarchi, H., et al. 1987. J Biol Chem. 262: 15840-4. PMID: 2445740
- Antidiabetic sulfonylureas control action potential properties in heart cells via high affinity receptors that are linked to ATP-dependent K+ channels. | Fosset, M., et al. 1988. J Biol Chem. 263: 7933-6. PMID: 2453509
- Glyburide blocks the relaxation response to BRL 34915 (cromakalim), minoxidil sulfate and diazoxide in vascular smooth muscle. | Winquist, RJ., et al. 1989. J Pharmacol Exp Ther. 248: 149-56. PMID: 2464055
- Mechanism of Action of Novel Glibenclamide Derivatives on Potassium and Calcium Channels for Insulin Secretion. | Frederico, MJS., et al. 2017. Curr Drug Targets. 18: 641-650. PMID: 27316908
- Glibenclamide Alleviates LPS-Induced Acute Lung Injury through NLRP3 Inflammasome Signaling Pathway. | Yang, J., et al. 2022. Mediators Inflamm. 2022: 8457010. PMID: 35185385
- A glibenclamide-sensitive TRPM4-mediated component of CA1 excitatory postsynaptic potentials appears in experimental autoimmune encephalomyelitis. | Fearey, BC., et al. 2022. Sci Rep. 12: 6000. PMID: 35397639
- Glibenclamide promoted functional recovery following sciatic nerve injury in male Wistar rats. | Ferdowsi, S., et al. 2022. Fundam Clin Pharmacol. 36: 966-975. PMID: 35524424
- Insulin-like and insulin-enhancing effects of the sulfonylurea glyburide on rat adipose glycogen synthase. | Altan, N., et al. 1985. Diabetes. 34: 281-6. PMID: 3918901
- Is there a concentration-effect relationship for sulphonylureas? | Melander, A., et al. 1998. Clin Pharmacokinet. 34: 181-8. PMID: 9533980
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Glyburide (Glibenclamide), 1 g | sc-200982 | 1 g | $46.00 | |||
Glyburide (Glibenclamide), 5 g | sc-200982A | 5 g | $61.00 | |||
Glyburide (Glibenclamide), 25 g | sc-200982D | 25 g | $117.00 | |||
Glyburide (Glibenclamide), 100 g | sc-200982B | 100 g | $173.00 | |||
Glyburide (Glibenclamide), 500 g | sc-200982C | 500 g | $530.00 |
