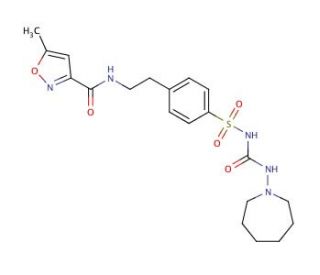
Glisoxepid (CAS 25046-79-1)
QUICK LINKS
Glisoxepid, a sulfonylurea derivative, plays a pivotal role in the field of biochemical research, particularly in studies focused on elucidating the mechanisms governing insulin secretion and glucose homeostasis. By targeting the ATP-sensitive potassium (K_ATP) channels on pancreatic beta cells, Glisoxepid facilitates the closing of these channels, leading to cellular depolarization. This depolarization triggers the opening of voltage-gated calcium channels, thereby enhancing the influx of calcium ions into the cells. The increase in intracellular calcium concentration is a signal that prompts the exocytosis of insulin-containing vesicles, thus boosting insulin release. This mechanism of action makes Glisoxepid an invaluable tool for researchers aiming to understand the intricate processes that regulate insulin release and its role in maintaining glucose levels within a physiological range. Additionally, the compound serves as a key reference molecule for comparative studies, aiding in the discovery and characterization of other compounds with potential applications in modulating insulin secretion.
Glisoxepid (CAS 25046-79-1) References
- [Therapy of type 2 diabetes. Critical evaluation of oral antidiabetic drugs]. | Landgraf, R. 2000. MMW Fortschr Med. 142: 33-7. PMID: 10872292
- Bepridil, an antiarrhythmic drug, opens mitochondrial KATP channels, blocks sarcolemmal KATP channels, and confers cardioprotection. | Sato, T., et al. 2006. J Pharmacol Exp Ther. 316: 182-8. PMID: 16174795
- Evidence for a saturable, energy-dependent and carrier-mediated uptake of oral antidiabetics into rat hepatocytes. | Petzinger, E. and Fückel, D. 1992. Eur J Pharmacol. 213: 381-91. PMID: 1618279
- Interaction of sulfonylureas with the transport of bile acids into hepatocytes. | Fückel, D. and Petzinger, E. 1992. Eur J Pharmacol. 213: 393-404. PMID: 1618280
- Simultaneous identification and validated quantification of 11 oral hypoglycaemic drugs in plasma by electrospray ionisation liquid chromatography-mass spectrometry. | Hess, C., et al. 2011. Anal Bioanal Chem. 400: 33-41. PMID: 21327875
- Repurposing existing drugs for new AMPK activators as a strategy to extend lifespan: a computer-aided drug discovery study. | Mofidifar, S., et al. 2018. Biogerontology. 19: 133-143. PMID: 29335817
- Second-Generation Antidiabetic Sulfonylureas Inhibit Candida albicans and Candidalysin-Mediated Activation of the NLRP3 Inflammasome. | Lowes, DJ., et al. 2020. Antimicrob Agents Chemother. 64: PMID: 31712208
- Identification of potential inhibitors of SARS-COV-2 endoribonuclease (EndoU) from FDA approved drugs: a drug repurposing approach to find therapeutics for COVID-19. | Chandra, A., et al. 2021. J Biomol Struct Dyn. 39: 4201-4211. PMID: 32462970
- The transport of L-cysteinesulfinate in rat liver mitochondria. | Palmieri, F., et al. 1979. Biochim Biophys Acta. 555: 531-46. PMID: 486467
- Inhibition of transglutaminase by hypoglycaemic sulphonylureas in pancreatic islets and its possible relevance to insulin release. | Gomis, R., et al. 1984. Res Commun Chem Pathol Pharmacol. 46: 331-49. PMID: 6151221
- Validity of the digitonin method for metabolite compartmentation in isolated hepatocytes. | Brocks, DG., et al. 1980. Biochem J. 188: 207-12. PMID: 7406879
- Effects of sulfonylureas on histochemical and ultracytochemical calcium distribution in B-cells of mice. | Klöppel, G. and Schäfer, HJ. 1976. Diabetologia. 12: 227-35. PMID: 782993
- The mitochondrial sulfonylurea receptor: identification and characterization. | Szewczyk, A., et al. 1997. Biochem Biophys Res Commun. 230: 611-5. PMID: 9015372
- Photohemolytic potency of oral antidiabetic drugs in vitro: effects of antioxidants and a nitrogen atmosphere. | Selvaag, E. 1996. Photodermatol Photoimmunol Photomed. 12: 166-70. PMID: 9017793
- Evaluation of phototoxic properties of oral antidiabetics and diuretics. Photohemolysis model as a screening method for recognizing potential photosensitizing drugs. | Selvaag, E. 1997. Arzneimittelforschung. 47: 1031-4. PMID: 9342417
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Glisoxepid, 10 mg | sc-391898 | 10 mg | $400.00 |
